Translate this page into:
Cytodiagnosis of Extra-nasal Rhinosporidiosis: A Study of 16 Cases from Endemic Area
Address for correspondence: Dr. Subrata Pal, E-mail: subratapal1985@gmail.com
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Context:
Extra-nasal rhinosporidiosis is not uncommon in endemic region like India. Clinical presentations of extra-nasal rhinosporidiosis lesion often lead to diagnostic dilemma. Cytology can help in the preoperative diagnosis of such lesions.
Aims:
The aims of our study were to find the clinico-pathological presentation of extra-nasal rhinosporidiosis and to evaluate the role of cytology in diagnosing these lesions preoperatively.
Settings and Design:
Fine-needle aspiration cytology is often used for preoperative diagnosis of sub-cutaneous lesions of the head and neck region. This retrospective study was designed to include the cytologically diagnosed cases of rhinosporidiosis and to compare with final histopathology of the lesions.
Materials and Methods:
A total of 21 cases of extra-nasal rhinosporidiosis were diagnosed in our study period of 18 months. Cytology was approached in 17 cases and 16 cases were diagnosed as rhinosporidiosis, which were included in the study group. Twelve cases were sampled by fine-needle aspiration and four cases by scrap technique. Histopathological confirmation was possible in all cytologically diagnosed cases.
Results:
Head and neck region were involved in 15 cases and only one case was on the skin of right upper arm. Orbital region was the most common extra-nasal site of involvement. Most of the cases (13 cases, 81.25%) belonged to the age group of 11-30 years. All cytologicaly diagnosed cases of rhinosporidiosis were concordant with histopathology. Only one false-negative case was cytologically diagnosed as suppurative inflammatory lesion. Sensitivity and specificity of cytology in diagnosis of extra-nasal rhinosporidiosis were 94.11% and 100% respectively.
Conclusions:
Extra-nasal rhinosporidiosis is an important differential diagnosis of nodular, polypoid mass of head-neck-face region. Cytology can be used as an important tool in preoperative diagnosis of extra-nasal rhinosporidiosis.
Keywords
Cytology
extra-nasal sites
rhinosporidiosis
Rhinosporidium seeberi
INTRODUCTION
Rhinosporidiosis is a chronic granulomatous disease of infective etiology. After the 1st case report from Argentina in 1900, it has been documented from about 70 countries from different geographical location.[12] India and Sri Lanka are the endemic regions for rhinosporidiosis due to temperate climate.[3] Rhinosporidiosis is caused by an organism of mesomycetozoea group known as Rhinosporidium seeberi.[1] Majority of the cases are sporadic but >90% of cases occur in South East Asia.[12] Polypoid nasal mass is the most common clinical manifestation of nasal rhinosporidiosis.[45] Extra-nasal (atypical) sites of rhinosporidiosis are orbital region, lip, palate, uvula, larynx, trachea, buccal cavity, lacrimal sac, scalp, skin, penis, urethra, vulva, and bone.[123] Presumptive diagnosis of primary rhinosporidiosis at extra-nasal site is often difficult. Some cases of extra-nasal rhinosporidiosis are associated with nasal involvement also. Few cases of disseminated rhinosporidiosis have also been reported. Extra-nasal rhinosporidiosis often presents with cutaneous or sub-cutaneous nodular swelling or sometimes with reddish polypoid mass lesion. In our study, we approached fine-needle aspiration and/or scraping of cutaneous or sub-cutaneous extra-nasal lesions for diagnostic purpose. The study was taken up with the objectives to observe the extra-nasal sites of rhinosporidiosis and to evaluate the role of fine-needle aspiration cytology (FNAC) as a diagnostic tool for these lesions.
MATERIALS AND METHODS
This study was conducted from May 2011 to December 2012 in the Department of Pathology of our college, a tertiary medical center situated in rural area of Eastern India. Ethical clearance was taken from Institutional Ethical Committee before starting the study. During the study period, 21 cases of extra-nasal rhinosporidiosis were diagnosed hisopathologically, among which 17 cases were undergone preoperative cytological evaluation. 16 cases were cytologically diagnosed as extra-nasal rhinosporidiosis and these were included in the study group. Four cases were not evaluated cytologically. Detailed clinical history about presenting features, age, sex, site of lesion, occupation, hygienic status, and other infections related with immunodeficiency was collected in all cases. Nasal rhinosporidiosis cases were not included in our study. After obtaining written consent, those patients were undergone aspiration cytology by standard 23-gauge needle attached with 10 ml syringe. In four cases, imprint cytology and scraping were done to avoid risk of bleeding. The smears were stained with May-GrunwaldGiemsa and Leishman-Giemsa stain and periodic acid-Schiff (PAS) stain. Cytological diagnosis was done by demonstration of diffusely scattered sporangia of varying sizes with transparent capsules containing sporangiospores admixed with nucleated squamous cells, cellular debris and inflammatory cells [Figures 1 and 2].
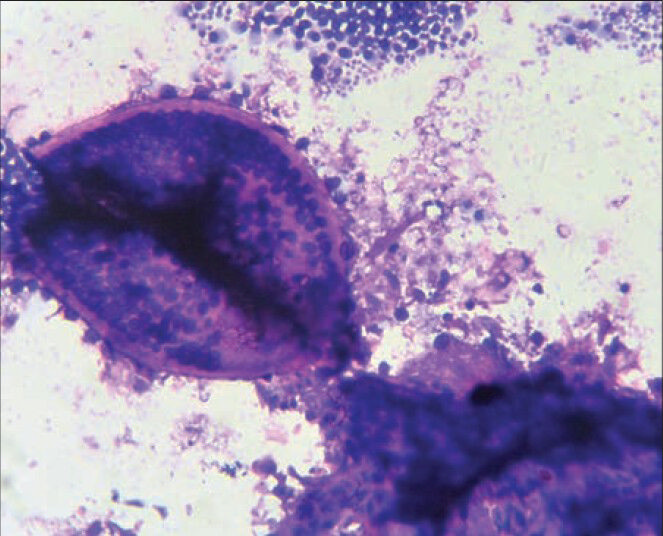
- Cytology smear of rhinosporidiosis showing clusters of endospores and intact sporangium (Leishman-Giemsa stain, ×400 view)
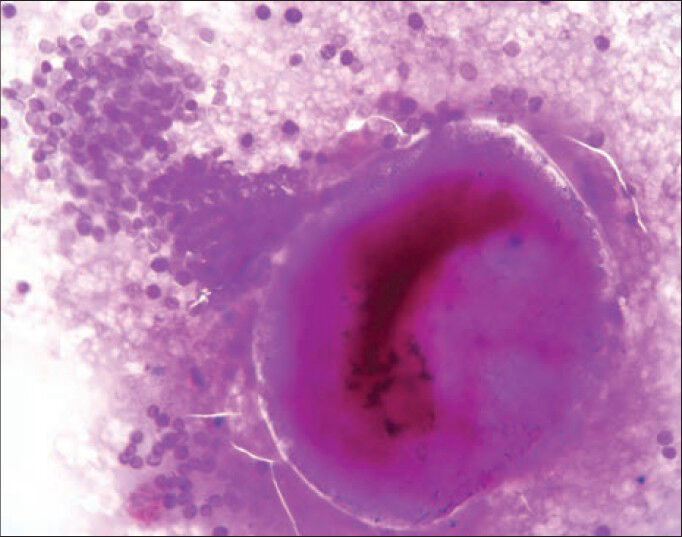
- Cytology smear of rhinosporidiosis showing a ruptured sporangium with many endospores and an intact sporangium (periodic acid-Schiff stain, ×400 view)
In all cases, lesions were excised and biopsy samples were processed as standard method of fixation, tissue processing and section cutting. Histopathological sections were stained with hematoxylin and eosin stain and PAS stain. Histopathological diagnosis was confirmed by demonstration of endospores and sporangia in developmental stages scattered in a fibrovascular stroma. Most of the cases showed moderate chronic inflammatory cell infiltration in stroma [Figures 3 and 4].
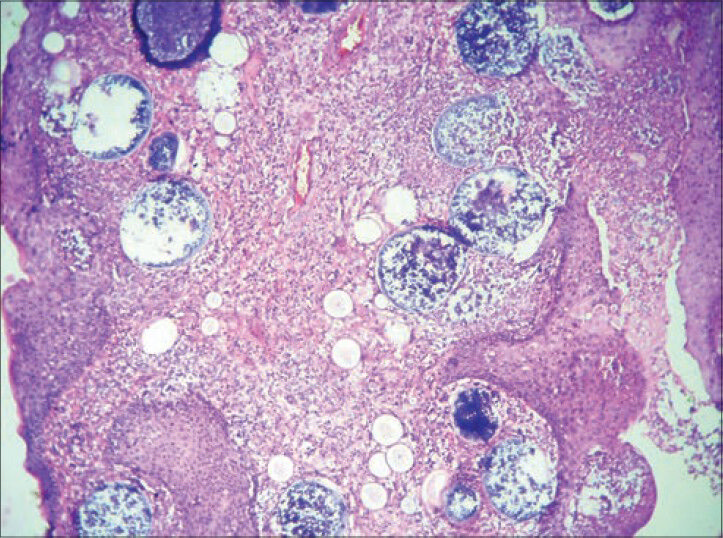
- Photomicrograph shows multiple variable sized sporangia with large number of microspores inside in histopathological examination of excised case (H and E, ×100 view)
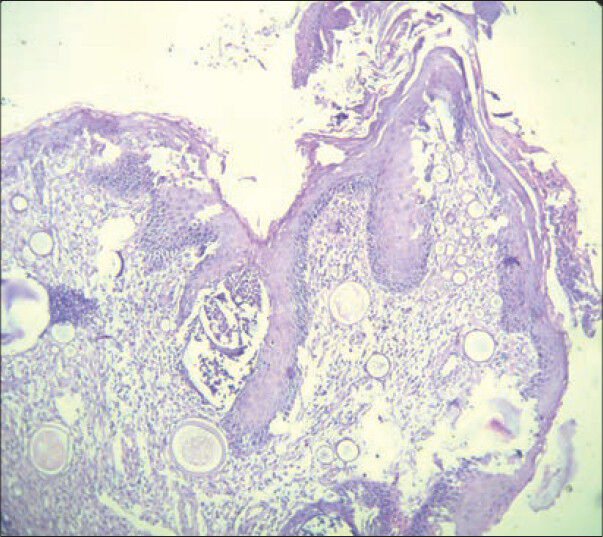
- Photomicrograph shows multiple variable sized sporangia in a histopathology section of a case of rhinosporidiosis (H and E, ×100 view)
RESULTS
Of 17 cases, we diagnosed 16 cases of rhinosporidiosis by fine-needle aspiration or scrap cytology involving different sites. Age range of the studied population had varied from 11 to 47 years. A large percentage of cases were among 11-30 years of age (13 cases; 81.25%). Among 16 cases, 10 cases (62.5%) were male and six cases (37.5%) were female. Most of the patients presented with swelling of head and neck region except one case, which was presented with swelling at right arm. Orbital tissue was the most common extra-nasal site in our study. Periorbital areas (eyelids, canthi, and nasolacrimal ducts) were involved in 8 (50%) cases. Nasolacrimal duct and lips were involved in two cases each. Root of the nose, parotid duct and scalp involvement were noted in single cases each. Most common presentation was lobular pedunculated soft to firm mass with irregular surface and sometimes superficial erosion. FNAC was done in 12 cases and scrape cytology was taken from four cases. All cases of cytologically diagnosed rhinosporidiosis were confirmed by biopsy and histopathological examination. Cytodiagnosis of all cases were concordant (100%) with histopathological findings.
Only one case of left eyebrow was cytologically misinterpreted as suppurative inflammatory lesion and subsequent biopsy and histopathology confirmed the case as rhinosporidiosis. Sensitivity of cytological diagnosis of extra-nasal rhinosporidiosis in the present study was 94.11% and specificity was approaching 100%.
DISCUSSION
Rhinosporidiosis was discovered by Malbran, as a sporozoon in 1892.[23] The same organism was first published by Seeber in 1900 and Ashworth describes its lifecycle in 1923.[6] Taxonomy of R. seeberi is still debatable and in last decade it was concluded that it is not a classic fungus, rather a human pathogen from Dermocystidium, the Rossette Agent, Ischthyophonus and Psorospermium clade - a clade of aquatic protistan parasites.[7] Rhinosporidiosis is more prevalent among males in the age group of 10-40 years.[3] Similarly, male patients were more frequent in our series (10 cases out of 16; 62.5%) and majority were young aged (13 cases are in the age group of 11-30 years; 81.25%). Mode of infection is supposed to be trans-epithelial inoculation of organism in traumatized epithelium.[12] Nasal cavity and nasopharynx are most common sites of rhinosporidiosis accounting 85% of the cases.[8] Extra-nasal rhinosporidiosis is not rare, especially in endemic areas. Ocular region (9%) is most common extra-nasal site of rhinosporidiosis.[8] It is also infrequently reported from different areas of skin and superficial epithelium. Extra-nasal rhinosporidiosis commonly present as sub-cutaneous lump or soft, friable, polypoid bleeding mass. Ocular rhinosporidiosis may manifest with watering, itching, conjunctivitis and photophobia.[9] Our cases also manifested similar findings. Nasal rhinosporidiosis is easier to diagnose clinically because of its typical polypoid presentation with a granular red surface with pinheaded spots. In contrast diagnosis of extra-nasal rhinosporidiosis is difficult on the basis of clinical presentation because it may be confused with benign cystic lesions, soft tissue tumors and papillomas. Hence, aspiration cytology will be helpful in preoperative diagnosis of these atypical presentations of rhinosporidiosis and for exclusion of differential diagnoses. Material for cytological diagnosis may be obtained by either fine-needle aspiration or scraping of superficially located lesions. Cytological diagnosis depends on demonstration of sporangia in different stages of maturation and endospores admixed with mixed inflammatory cells [Figures 1 and 2]. Sporules may be present as well-circumscribed round structures with several endospores inside [Figure 1].[10] Sometimes epithelial cells may be confused with endospores.
We have used PAS stain in three cases aspirated from nasolacrimal area, because of difficulty in separating endospores from nucleated respiratory epithelial cells as per previous authors recommendation.[911] PAS stain distinguished these by staining endospores (“comet” as referred by Beattie)[12] magenta colored where epithelial cells are PAS negative.[14]
We have misinterpreted only one case as suppurative inflammatory lesion in cytology smear. The aspirate was pus like and the smears revealed plenty of neutrophils, macrophages, and nuclear remnants in a necrotic background. No spore and capsular structure were identified. Secondary bacterial infection and supper added inflammation possibly caused abscess formation and improper site of needling produced cytology of inflammatory lesion in this case.
Definitive diagnosis of rhinosporidiosis depends on the histopathological examination of resected specimen. Histopathology reveals different stages of maturing sporangia, enclosed by thin chitinous walls and contain numerous endospores inside [Figures 3 and 4]. Surrounding tissue is loose and fibro vascular stroma is infiltrated with mixed inflammatory cells (lymphocytes, plasma cells, macrophages and neutrophils). Unlike other invasive mycoses, eosinophilic infiltration is absent as described by previous workers.[13]
Rhinosporidiosis lesions may be confused with other fungal lesions in cytology as well as histology. It should be differentiated from myospherulosis (sub-cutaneous spherulocytic disease), Coccidioides immitis and Chrysosporium parvum var. crescens.[2] Special stain like Gomori methanamine silver, Gridley's and PAS stain, mucicarmine stains help to distinguish them from other organisms.[12] C. immitis have similar thick spherical wall with endospores inside, but sporules are smaller (20-80 μm vs. 50-100 μm in rhinosporidiosis) and arthoconidia and hyphae may be present in C. immitis lesions.[14] Endospores of R. seeberi are longer in size and numerous in contrast to C. immitis.[12]
Definitive management includes surgical excision combined with electro-coagulation. Antifungal and bacterial drugs are not effective in R. seeberi lesions. Only drug that is used to prevent recurrence is dapsone.
In conclusion, Extra-nasal rhinosporidiosis is uncommon disease and often difficult to diagnose on clinical examination. Cytology can easily differentiate from other differential diagnosis like benign cystic lesions (epidermal inclusion cysts), soft tissue lesions and papillomas and helps in preoperative diagnosis. Until now, histopathology is gold standard for diagnosis of rhinosporidiosis but cytology has a great role in diagnosis of unsuspected extra-nasal lesions.
ACKNOWLEDGMENT
We want to acknowledge Prof. (Dr.) Swapan Pathak for his kind co-operation.
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- Clinicopathological study of rhinosporidiosis with special reference to cytodiagnosis. J Cytol. 2012;29:246-9.
- [Google Scholar]
- Recent advances in rhinosporidiosis and Rhinosporidium seeberi. Indian J Med Microbiol. 2002;20:119-31.
- [Google Scholar]
- Atypical presentations of rhinosporidiosis: A clinical dilemma? Indian J Otolaryngol Head Neck Surg. 2011;63:243-6.
- [Google Scholar]
- Rhinosporidiosis of lacrimal sac: A rare case report from North East India. Indian J Med Sci. 2011;65:40-2.
- [Google Scholar]
- Nasal rhinosporidiosis-four cases relate literature review. Int Arch Otorhinolaryngol. 2007;11:428-9.
- [Google Scholar]
- On Rhinosporidium seeberi (Wernicke, 1903) with special reference to its sporulation and affinities. Trans R Soc Edinb. 1923;53:301-42.
- [Google Scholar]
- Phylogenetic analysis of Rhinosporidium seeberi's 18S small-subunit ribosomal DNA groups this pathogen among members of the protoctistan Mesomycetozoa clade. J Clin Microbiol. 1999;37:2750-4.
- [Google Scholar]
- Ocular rhinosporidiosis with staphyloma formation: A case with unusual features. Eye Brain. 2011;3:1-4.
- [Google Scholar]
- Cytologic and differential diagnosis of rhinosporidiosis. Acta Cytol. 1994;38:361-6.
- [Google Scholar]
- Disseminated cutaneous rhinosporidiosis: Diagnosis by fine needle aspiration cytology. Acta Cytol. 2006;50:111-2.
- [Google Scholar]
- Rhinosporidium kinealyi: a sporozoan of the nasal mucous membrane. J Path Bact. 1906;11:270-5.
- [Google Scholar]
- The humoral immune response in human rhinosporidiosis. Proc Kandy Soc Med. 1999;21:9.
- [Google Scholar]
- Rhinosporidiosis: Intraoperative cytological diagnosis in an unsuspected lesion. Case Rep Pathol 2012 2012:101832.
- [Google Scholar]





