Translate this page into:
Activity of cefotaxime versus ceftriaxone against pathogens isolated from various systemic infections: A prospective, multicenter, comparative, in vitro Indian study
*Corresponding author: Ajitkumar A. Gondane, Medical Affairs, Alkem Laboratories, Devashish Building, Alkem House, Senapati Bapat Road, Lower Parel, Mumbai 400013, Maharashtra, India ajitkumar.gondane@alkem.com
-
Received: ,
Accepted: ,
How to cite this article: Gondane AA and Pawar DB. Activity of cefotaxime versus ceftriaxone against pathogens isolated from various systemic infections: A prospective, multicenter, comparative, in vitro Indian study. J Lab Physicians. 2024;16:97-104. doi: 10.1055/s-0043-1772564
Abstract
Objectives:
To determine the susceptibility of isolated pathogens with different samples collected from patients taking cefotaxime as compared with ceftriaxone.
Materials and Methods
In vitro susceptibility study was conducted at microbiology laboratories of east (Bhubaneshwar), west (Ahmedabad), north (Delhi), and south (Srikakulam) India. Samples of treatment naïve patients with various clinical infections were included if they were positive for bacterial culture. Minimum inhibitory concentration (MIC) and zone of inhibitions (ZoIs) for each isolate were determined using Ezy MIC strip test and disk diffusion methods, respectively. Findings of MIC and ZoI were interpreted as per the Clinical and Laboratory Standards Institute guidelines. Appropriate statistical tests were used.
Statistical Analysis:
Probable diagnosis, type of sample, and isolated organisms was presented as the proportion. Geometric mean was calculated for MIC and arithmetic mean was calculated for ZoI and they were presented as mean (95% confidence interval).
Results:
Four hundred clinical samples of urinary tract infection (42.75%), lower respiratory tract infection (20.75%), skin and soft tissue infection (16.75%), and sepsis (12.75%) were evaluated. Escherichia coli (47.75%) was the most common organism isolated followed by Klebsiella (26%), Salmonella (7.75%), and Proteus mirabilis (3.75%). The mean MIC values for E. coli, Klebsiella, Staphylococcus, Citrobacter koseri, and Serratia marcescens were found to be lower when treated with cefotaxime compared with ceftriaxone, although the difference was not statistically significant. However, cefotaxime produced significantly (p < 0.05) more ZoI for E. coli, Klebsiella, and Salmonella as compared with ceftriaxone.
Conclusion:
Cefotaxime has shown better sensitivity profile in terms of MIC and ZoI to most of the isolated organisms as compared with ceftriaxone and thus can be preferred for empirical treatment of such patients.
Keywords
antimicrobial
resistance
cephalosporins
strip test
INTRODUCTION
Cefotaxime, a potent semisynthetic cephalosporin, exhibits broad-spectrum antimicrobial activity against a variety of gram-positive organisms, including methicillin-sensitive Staphylococcus aureus, groups A and B streptococci (including Streptococcus pyogenes), β-hemolytic streptococci (groups C, F, G), and viridans group streptococci. It also shows efficacy against gram-negative aerobic bacteria such as Haemophilus influenzae and parainfluenzae, Neisseria gonorrhoeae, Moraxella catarrhalis, Neisseria meningitidis, Proteus spp., Providencia spp., Yersinia enterocolitica, as well as certain anaerobic bacteria.[1-3] As a third-generation cephalosporin, cefotaxime generally demonstrates greater activity against gram-negative bacteria compared with first and second-generation cephalosporins. It inhibits bacterial cell wall synthesis by interfering with the transpeptidation during the peptidoglycan synthesis. 3-lactamase stability (types III, IV, V, and I); good permeability through the cell membrane; strong affinity for lethal penicillin-binding proteins la, lb(s), and III; minimal limitation by the inoculum effect; and bactericidal action at or close to the inhibitory concentration are the various characteristics which make the cefotaxime a highly potent drug.
Cefotaxime is the preferred choice for treating various infections, including lower respiratory tract infections (LRTIs), urinary tract infections (UTIs), skin and soft tissue infections, and genital infections where penicillin is ineffective or unsuitable. It is also recommended for intra-abdominal infections, including peritonitis, as well as for the treatment of acute meningitis, sepsis, and endocarditis when used in combination with other appropriate antibiotics.[3] Cefotaxime has become the most extensively utilized antibiotic due to its broad-spectrum effectiveness and its inclusion as the recommended empiric (presumptive) or first-line therapy for a diverse range of severe infections in the National Treatment Guidelines for Antimicrobial Use in Infectious Diseases, issued by the Government of India.[4]
Ceftriaxone is another third-generation cephalosporin which shares similar characteristics to cefotaxime except for serum protein binding (95 vs. 35%) and elimination halflife (8.8 vs. 1.2 hours).[5] Serum protein binding may diminish therapeutic efficacy by affecting antibacterial activity and drug distribution. In an in vitro study conducted by Nath et al, it was concluded that the in vitro antimicrobial activity of ceftriaxone compared with that of cefotaxime was significantly diminished in the presence of serum albumin.[5] Thus, high serum protein binding of ceftriaxone may limit its in vivo activity. Ceftriaxone is indicated for the same conditions as cefotaxime, demonstrating overlapping areas of use.
Antibiotic therapy plays a fundamental role in treating infectious diseases. It is crucial to establish the diagnosis of an infectious disease by determining the site of infection and conducting microbiological testing to identify the causative agent and its susceptibility to specific antibiotics before initiating any antibiotic treatment. In order to ensure accurate diagnosis and appropriate treatment, health care providers should collect diagnostic specimens in a proper manner and promptly submit them to the laboratory before initiating antibiotic therapy. Since microbiological results typically take 24 to 72 hours to become available, initial treatment for infections is often started empirically based on the clinical presentation to prevent deterioration or complications. Hence, it is imperative for the clinician to know the sensitivity pattern of the common causative organisms circulated in their local environment to help them decide the choice of antibiotic for empiric treatment of infections. As both of these antibiotics are used in severe life-threatening infections, it becomes extremely important for the clinicians to know its current sensitivity pattern against the organisms in Indian region. The reports of resistance to cefotaxime and ceftriaxone have become apparent which warrants antibiotic susceptibility testing. Antibiotic susceptibility of microorganisms can be tested by various methods such as disc diffusion method, microdilution technique, or the strip method. Conventional methods have many disadvantages, for example, they are tedious, require manual undertaking, and have risk of errors in the preparation of antimicrobial solutions for each test. Strip method is the best technique to be used when only few antibiotics are to be tested or in case of fastidious organism. In addition, the strip method offers the advantage of providing easily reproducible and quantitative wide range minimum inhibitory concentrations (MICs).[6] The current study was planned to evaluate the susceptibility of clinical bacterial isolates to cefotaxime obtained from different samples of patients suffering from common clinical infectious conditions and compared them with ceftriaxone by in vitro strip and disc methods.
MATERIALS AND METHODS
Study setting
The present prospective, active controlled, in vitro study was conducted on different biological samples received in four microbiology laboratories for routine clinical testing from patients suffering from various infections wherein cefotaxime is routinely indicated. The study encompassed microbiology laboratories across different regions of India, including Bhubaneshwar in the east, Ahmedabad in the west, Delhi in the north, and Srikakulam in the south. Ethics committees of respective sites reviewed and approved the study protocol and corresponding documents. Samples of treatment naïve patients suffering from infections of the lower respiratory tract, urinary tract, skin, soft tissues; intra-abdominal infections (including peritonitis); genital infections caused by gonococci; acute meningitis, sepsis, and endocarditis were included if they were positive for bacterial culture. Patients who had a documented history of receiving antibiotics, antiviral agents, or interferon therapy within the last 5 days were excluded from the study. Samples that met the predefined inclusion and exclusion criteria were subjected to testing for antibiotic sensitivity of cefotaxime and ceftriaxone using strip and disc methods. This allowed for the determination of MIC and zone of inhibition (ZoI) for the respective antibiotics. Details of probable diagnosis were collected for each included sample. Ezy MIC strips and discs manufactured by HiMedia Laboratories, India were used to determine MIC and ZoI.
Culture and identification of bacteria
Culture media and inoculum were prepared as per standard methods. Each sample was inoculated on McConkey agar, chocolate agar, and blood agar. The inoculum on the plate was streaked out for discrete colonies with a sterile wire loop.
After inoculation, the culture plates were placed in an incubator at a temperature of 37°C for a duration of 24 hours. During this incubation period, the plates were regularly monitored for the growth of bacterial colonies. The isolated bacteria were subsequently subjected to morphological and biochemical tests, following the standardized procedure outlined by Cheesbrough.[7] Positive samples were subjected to susceptibility testing by strips and discs methods.
Mic determination by strip test method
Ezy MIC strips is a quantitative technique that determines the MIC in μg/mL and antibiotic susceptibility of various aerobic and fastidious organisms using the concepts of dilution and diffusion principles for susceptibility testing. It directly quantifies antibiotic susceptibility in terms of discrete MIC values, which are more precise and reproducible as compared with conventional procedures. Ezy MIC strip is a thin, inert, and porous paper strip coated with antibiotic that has MIC reading scale in μg/mL over both sides of the strip. It also contains two or three-letter symbol printed on the top side, which represents the antibiotic. The antibiotic is dried and stabilized as a predefined exponential gradient with maximum concentration at one end and minimum at the other end, which covers a continuous concentration range across 15 twofold dilutions such as conventional method. There is gradual and effective transfer of the preformed antibiotic gradient from the strip into the agar medium when strip is applied to the medium. A stable, continuous, and exponential gradient of antibiotic concentrations is formed directly underneath the strip. The bacterial growth inhibition appeared as elliptical zones centered along the Ezy MIC strip, allowing for the determination of MIC values. The MICs were read from a scale printed on the strip, which covered a range of 0.016 to 256 μg/mL. Cefotaxime (test) and ceftriaxone (control) Ezy MIC strips manufactured by HiMedia that can measure MICs from 0.016 to 256 μg/mL were used in the study. The strips were placed on the plate using the applicator as per standard laboratory practice. Once positioned, the strips were not repositioned or adjusted, ensuring complete contact with the agar surface. The plates were then allowed to dry for 10 to 15 minutes and incubated them inverted under appropriate conditions. Readings were taken only when sufficient bacterial growth was observed. When growth occurred along the entire strip, MIC was read as > the highest values on the MIC strip, whereas in case of the inhibition ellipse is below the strip (did not intersect the strip), MIC was read as < the lowest value on the MIC scale.
Zone of inhibition using disk diffusion method
The disk diffusion method was used as described by Bauer et al.[8] A 6-mm filter paper disk impregnated with a known concentration of an antimicrobial compound was placed on the agar plate. Complete contact with the agar surface of the disks was confirmed and agar plates were dried for 10 to 15 minutes. They were then incubated in an inverted position with appropriate conditions. When antibiotic concentration inhibits the growth and organism is sensitive, it forms a clear zone around the specific disc and thus creates a ZoI.
To assess the sensitivity of bacteria to antibiotics, the diameter of the ZoI was measured in millimeter (mm) using a caliper. The interpretation of the ZoI was done according to the guidelines provided by the Clinical and Laboratory Standards Institute (CLSI). For this purpose, antibiotic discs of cefotaxime and ceftriaxone, manufactured by HiMedia, were utilized to measure the ZoI.
ASSESSMENT OF EFFICACY
Determining MIC50
The MIC50 represented the value at which ≥50% of the isolates in a test population were inhibited. Given “n” test strains and the values y1, y2…yn were representing a graded series of MICs starting with the lowest value, the MIC50 was the value at position n × 0.5, when “n” was an even number of test strains and (n + 1) × 0.5 if “n” was an odd number of test strains.
INTERPRETATION OF RESULTS AS PER CLSI GUIDELINES
The MIC results and the ZoIs obtained for the cefotaxime and ceftriaxone were evaluated as per CLSI (M100 2022) guidelines and classified as sensitive/resistant (intermediate was considered as resistant for the study for available strains based on the MIC values and ZoI.[9]
Statistical analysis
Probable diagnosis, type of sample, and isolated organisms was presented as the proportion. Geometric mean was calculated for MIC and arithmetic mean was calculated for ZoI and they were presented as mean (95% confidence interval). The comparisons between MIC and ZoI between cefotaxime and ceftriaxone were performed using unpaired t test, whereas proportions of sensitive organisms were compared using chi-square test. A p < 0.05 was considered as a significant difference.
RESULTS
Total 400 samples were evaluated. The most common probable diagnosis was UTI (42.75%) followed by LRTI (20.75%), skin and soft tissue infection (16.75%), and sepsis (12.75%) (Figure 1). Urine (42.75%) was the most common sample followed by pus (17.5%), sputum (15.75%), blood (12.75%), endotracheal secretion (5%), and endocervical secretion (3.75%) (Figure 2). Among the 400 culture positive samples subjected to antibiotic sensitivity evaluation during the study, E. coli (47.75%) was the most common organism isolated followed by Klebsiella (26%), Salmonella (7.75%), and Proteus mirabilis (3.75%) (Figure 3).
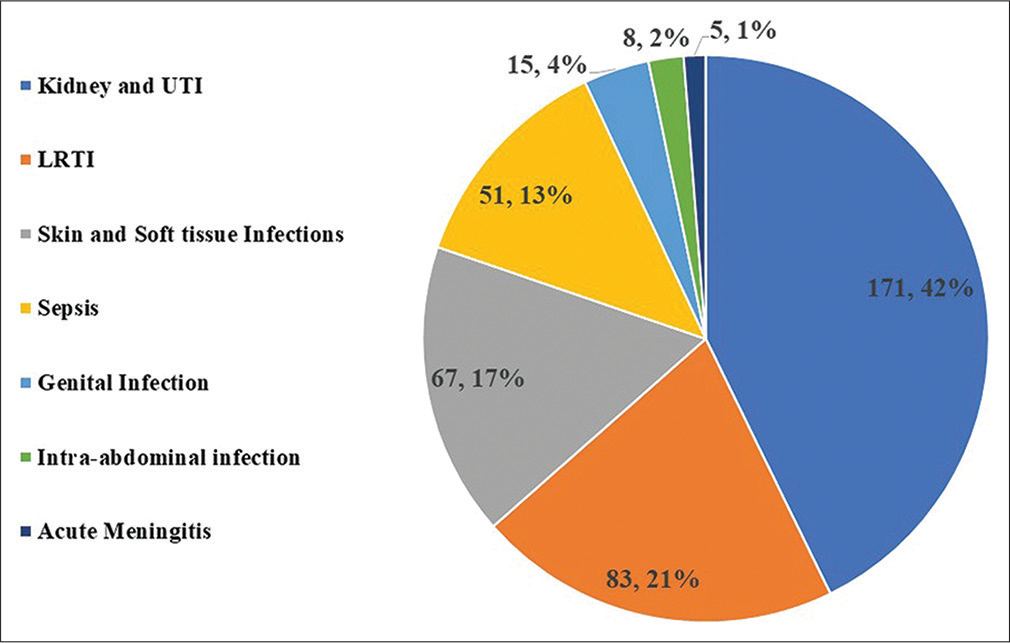
- Probable diagnosis of patients (values are in numbers, percentage). LRTI, lower respiratory tract infection; UTI, urinary tract infection.
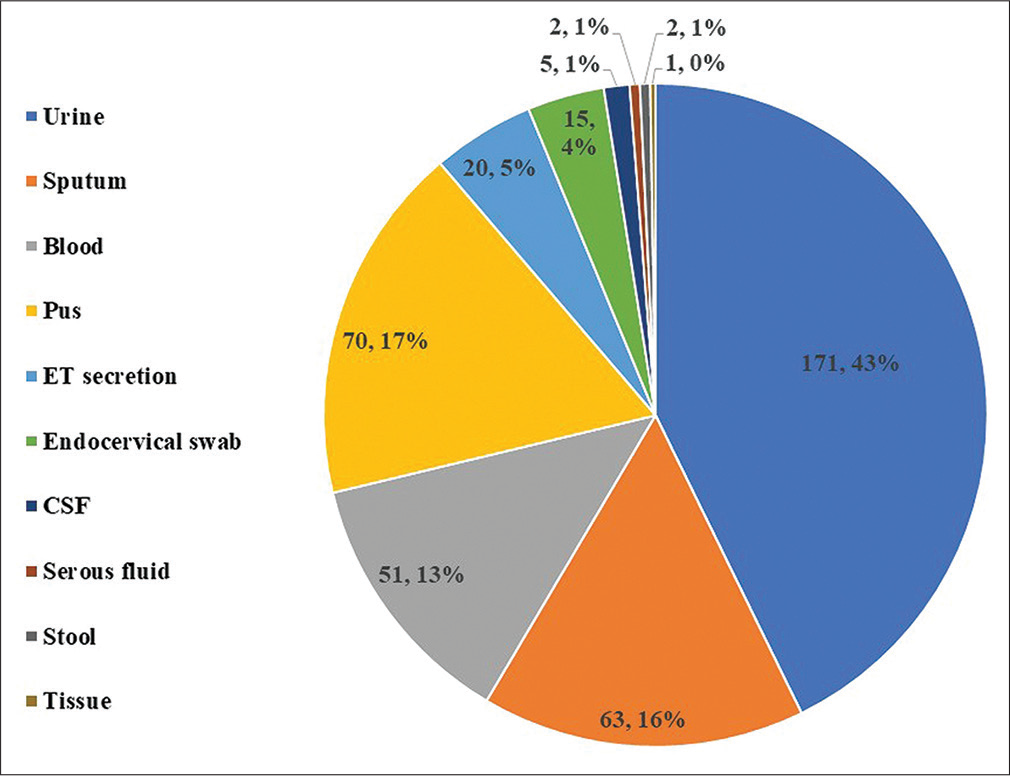
- Type of samples analyzed in the study (values are in numbers, percentage).
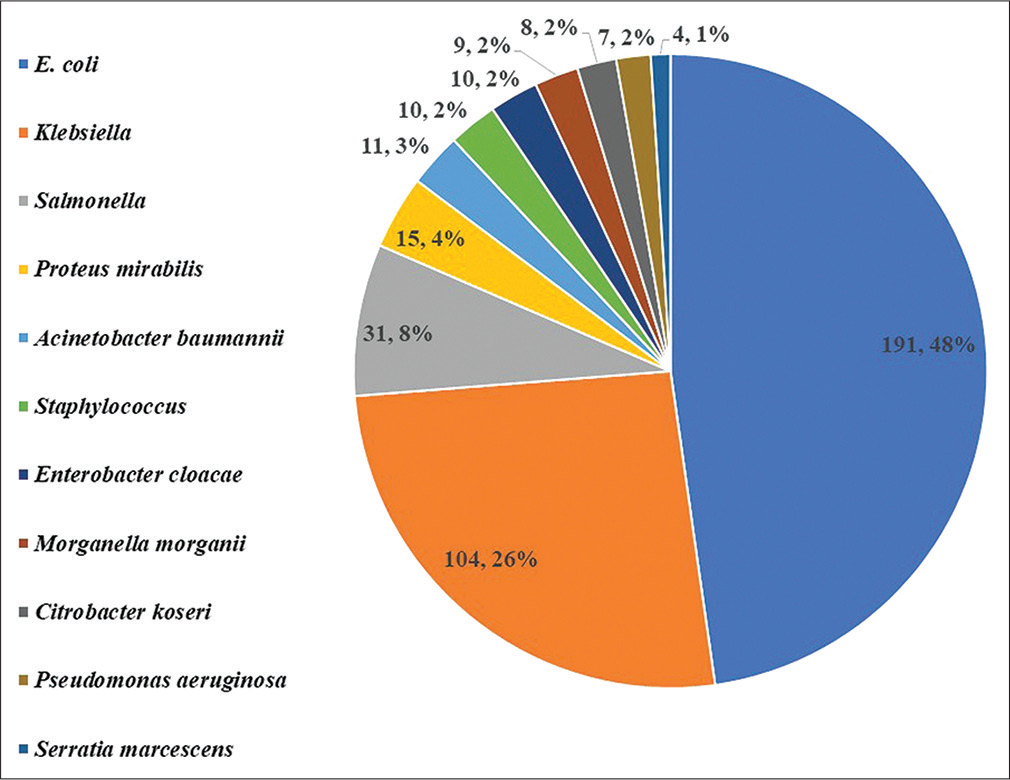
- Distribution of isolated organisms from culture (values are in numbers, percentage).
EFFICACY ANALYSIS
As shown in Table 1, mean MIC for E. coli, Klebsiella, Staphylococcus, Citrobacter koseri, and Serratia marcescens were lower for cefotaxime as compared with ceftriaxone, although not statistically significant. However, cefotaxime produced significantly (p < 0.05) more ZoI for E. coli, Klebsiella, and Salmonella as compared with ceftriaxone (Table 2).
| Organisms | Cefotaxime | Ceftriaxone | p-Value |
|---|---|---|---|
| E. coli (n = 191) | 1.02 (0.64–1.64) | 1.26 (0.79–2.01) | 0.54 |
| Klebsiella (n = 104) | 0.70 (0.40–1.21) | 0.95 (0.56–1.63) | 0.42 |
| Salmonella (n = 31) | 0.08 (0.05–0.13) | 0.08 (0.06–0.13) | 0.82 |
| Proteus mirabilis (n = 15) | 0.11 (0.02–0.45) | 0.10 (0.02–0.45) | 0.98 |
| Acinetobacter baumannii (n = 11) | 43.43 (6.6–286.2) | 57.36 (8.5–388.1) | 0.82 |
| Staphylococcus (n = 10) | 0.75 (0.11–5.0) | 0.92 (0.13–6.3) | 0.86 |
| Enterobacter cloacae (n = 10) | 5.36 (0.39–74.1) | 9.7 (1.07–88.3) | 0.70 |
| Morganella morganii (n = 9) | 0.54 (0.10–2.9) | 0.43 (0.07–2.81) | 0.84 |
| Citrobacter koseri (n = 8) | 0.31 (0.02–4.4) | 0.35 (0.02–5.7) | 0.94 |
| Pseudomonas aeruginosa (n = 7) | 92.8 (27.6–312.4) | 42.4 (2.6–695.2) | 0.54 |
| Serratia marcescens (n = 4) | 0.09 (0.06–0.14) | 0.12 (0.06–0.26) | 0.37 |
Abbreviation: MIC, minimum inhibitory concentration.
Note: Data presented as geometric mean (95% confidence interval) of MIC.
p-Value based on unpaired t test of log transformed data.
| Organism | Cefotaxime | Ceftriaxone | p-Value |
|---|---|---|---|
| E. coli (n = 191) | 24.1 (22.7–25.4) | 22.1 (20.1–23.3) | 0.03a |
| Klebsiella (n = 104) | 24.2 (22.5–25.8) | 21.7 (20.3–23.2) | 0.03a |
| Salmonella (n = 31) | 29.1 (27.6–30.5) | 26.5 (24.8–28.2) | 0.02a |
| Proteus mirabilis (n = 15) | 28.3 (24.8–31.9) | 28.2 (24.2–32.3) | 0.95 |
| Acinetobacter baumannii (n = 11) | 14.5 (8.4–20.6) | 12.8 (6.9–18.7) | 0.67 |
| Staphylococcus (n = 10) | 24.0 (19.8–28.2) | 22.1 (18.9–25.3) | 0.43 |
| Enterobacter cloacae (n = 10) | 17.8 (10.1–25.6) | 16.8 (9.6–24.0) | 0.83 |
| Morganella morganii (n = 9) | 25.7 (21.8–29.5) | 24.6 (20.4–28.7) | 0.66 |
| Citrobacter koseri (n = 8) | 24.6 (18.7–30.6) | 23.1 (17.6–28.7) | 0.67 |
| Pseudomonas aeruginosa (n = 7) | 17.6 (12.1–23.0) | 17.0 (10.7–23.3) | 0.87 |
| Serratia marcescens (n = 4) | 28.5 (25.7–31.3) | 26.8 (25.2–28.3) | 0.13 |
Note: Data presented as mean (95% confidence interval) of zone of inhibition.
a p<0.05 for unpaired t test is showing statistical significance.
MIC50 values for each organism are shown in Table 3 for both cefotaxime and ceftriaxone.
| Organism | Cefotaxime | Ceftriaxone |
|---|---|---|
| E. coli (n = 191) | 0.5 | 0.5 |
| Klebsiella (n = 104) | 0.38 | 0.5 |
| Salmonella (n = 31) | 0.064 | 0.064 |
| Proteus mirabilis (n = 15) | 0.032 | 0.047 |
| Acinetobacter baumannii (n = 11) | 192 | 256 |
| Staphylococcus (n = 10) | 0.125 | 0.125 |
| Enterobacter cloacae (n = 10) | 4 | 4 |
| Morganella morganii (n = 9) | 0.19 | 0.25 |
| Citrobacter koseri (n = 8) | 0.047 | 0.047 |
| Pseudomonas aeruginosa (n = 7) | 256 | 256 |
| Serratia marcescens (n = 4) | 0.064 | 0.094 |
Abbreviation: MIC, minimum inhibitory concentration.
Note: Values represent the MIC at which ≤50% of the strains within a test population are inhibited.
Based on MIC and ZoI cutoff levels defined in CLSI guidelines, more proportions of E. coli, and Klebsiella were found to be sensitive to cefotaxime as compared with ceftriaxone, but it could not achieve the statistical significance. The sensitivity pattern of all organisms against cefotaxime and ceftriaxone as interpreted based on MIC and ZoI is shown in Figures 4 and 5, respectively.
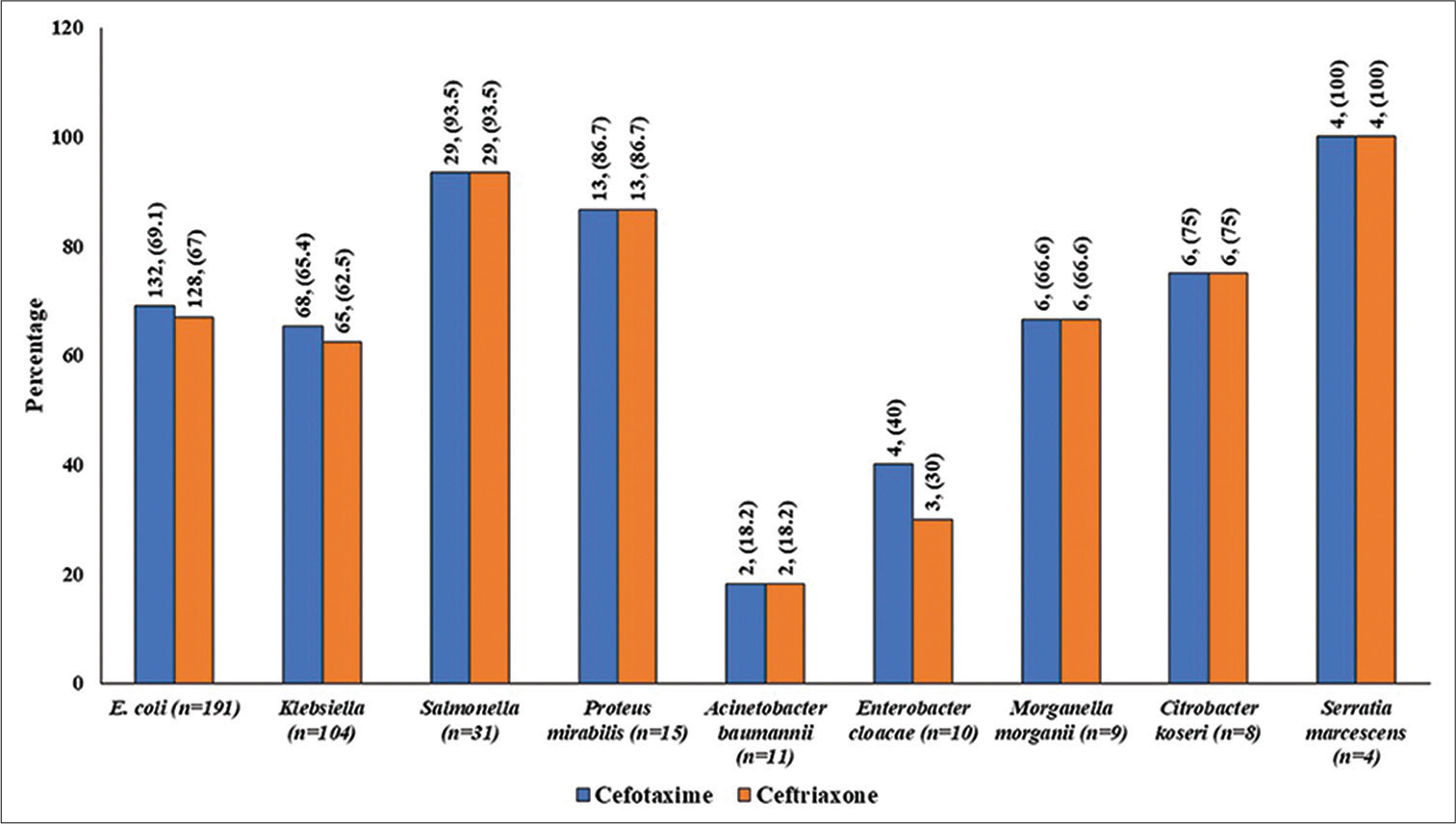
- Proportion of samples sensitive for cefotaxime versus ceftriaxone as per CLSI criteria based on MIC (values are in numbers, percentage). CLSI, clinical and Laboratory Standards Institute; MIC, minimum inhibitory concentration.
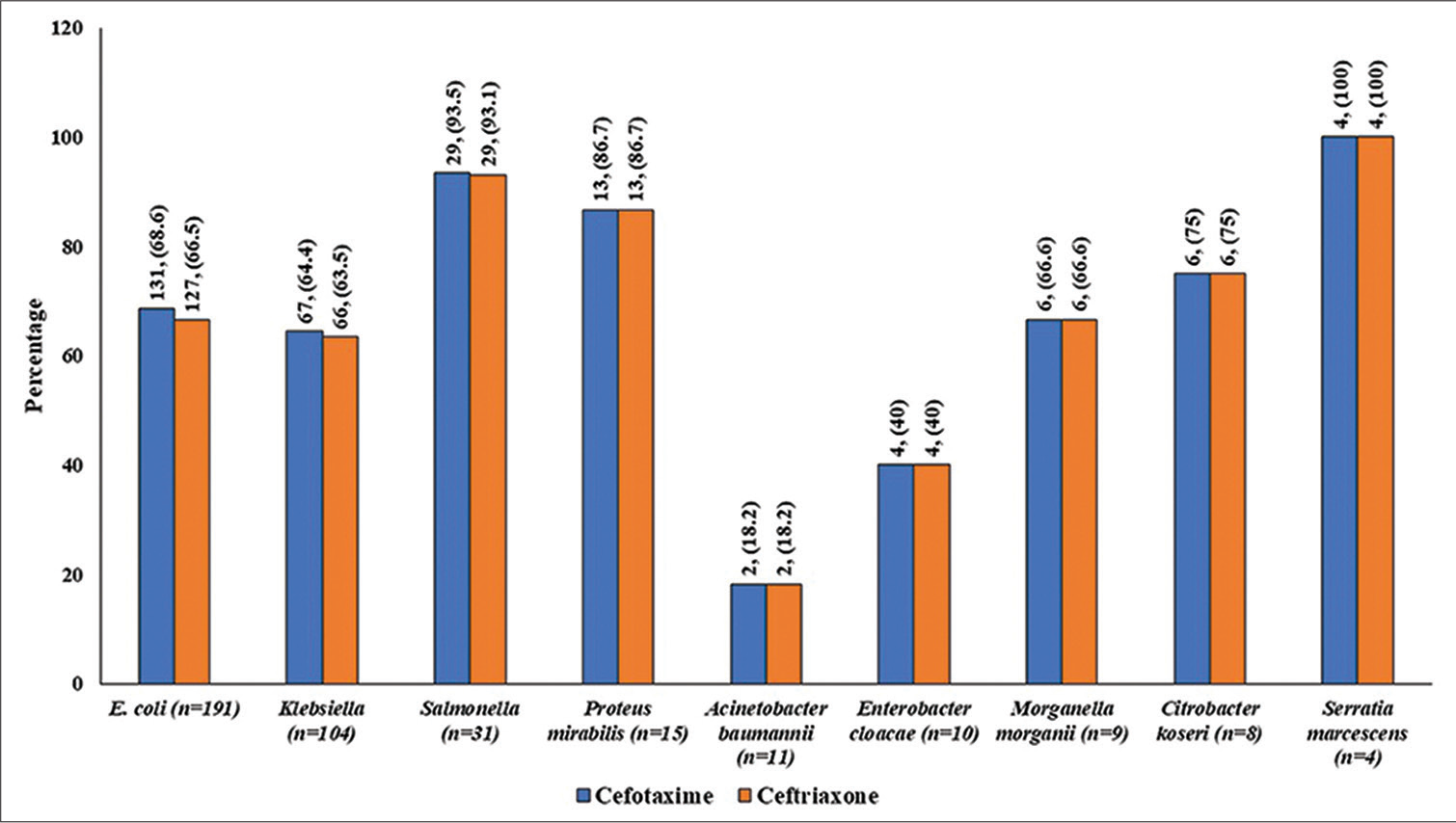
- Proportion of samples sensitive for cefotaxime versus ceftriaxone as per CLSI criteria based on zone of inhibition (values are in numbers, percentage). CLSI, clinical and Laboratory Standards Institute.
DISCUSSION
The current prospective, multicenter, in vitro study compared the sensitivity pattern between cefotaxime and ceftriaxone (third-generation cephalosporins) in India for various organisms isolated from clinical samples of patients indicated for treatment with cefotaxime/ceftriaxone. Up to the best of our knowledge, recent data on susceptible and resistant organisms from clinical isolates for cefotaxime and ceftriaxone are scarce from Indian region.
In this study, authors found that the mean MIC for E. coli, Klebsiella, Staphylococcus, C. koseri, and S. marcescens were lower for cefotaxime as compared with ceftriaxone, although not statistically significant. In addition, cefotaxime produced significantly (p < 0.05) more ZoI for E. coli, Klebsiella, and Aalmonella as compared with ceftriaxone. In the study conducted by Asari et al in 1983, cefotaxime showed higher antibacterial activity as compared with others for S. pneumoniae, S. pyogenes, S. agalactiae, E. coli, indole-positive Proteus, P. mirabilis, Klebsiella sp., S. marcescens, H. influenzae, and E. cloacae.[10] Similar trends were found with cefotaxime when susceptibility of strains (S. aureus, S. pneumoniae, S. agalactiae, S. pyogenes, E. coli, E. cloacae, and P. morganii) from clinical isolates was compared with latamoxef, cefoperazone, and ceftizoxime.[11,12] In recent study conducted in Bangladesh on uropathogens, 57% E. coli, 80% Klebsiella spp., 50% Enterobacter spp., 71% Proteus, and 57% Pseudomonas were found sensitive to cefotaxime.[13]
Ceftriaxone and cefotaxime have similar antimicrobial spectrum and susceptibility of destruction to beta-lactamases, but have different serum protein binding capacity and elimination half-life. Ceftriaxone is 95% protein bound and its elimination half-life is 8.8 hours, whereas cefotaxime is less protein bound (35%) with elimination half-life of hours.[5] Serum protein binding may inhibit the therapeutic efficacy by altering antibacterial activity and drug distribution. Thus, high serum protein binding of ceftriaxone may in fact limit its in vivo activity.[14] The relationships between serum protein binding and the distribution of beta-lactam antibiotics have been extensively studied. In a study conducted by Nath et al, it is demonstrated that the bactericidal activity (in terms of survival rate of bacteria when exposed to a drug in broth containing 48 and 95% equivalent concentrations of serum albumin) of ceftriaxone was significantly lower as compared with cefotaxime (p < 0.001).[5] In study conducted in Rwanda, the resistance pattern to cefotaxime and ceftriaxone was similar for hospital and community-acquired infections.[15] In in vitro study conducted long back on major four species causing meningitis, MIC90, and MBC90 values were less for cefotaxime than ceftriaxone, and cefotaxime was found superior in terms of MBC90.[16] Clinically, cefotaxime and ceftriaxone show the comparable efficacy for various clinical conditions.[17,18]
Various studies have also compared the changes in bacterial flora and adverse effects between cefotaxime and ceftriaxone. In a study by Bräutigam et al, differencebetweenpostoperative infection rates between cefotaxime and ceftriaxone was nonsignificant, but significant increase of Candida species in vaginal flora after 24 and 48 hours was noted in ceftriaxone group.[19] They also found changes in thebowel flora, which was consistent with findings of other studies. Guggenbichler et al compared the impact of cefotaxime and ceftriaxone on the fecal microflora of pediatric patients. They observed that the first dose of ceftriaxone eradicated the susceptible enteric bacteria within 48 hours, leading to an overgrowth of Candida spp. and enterococci. Cefotaxime, however, had no essential suppressive effect.[20] Additionally, many previous studies have reported high incidences of diarrhea with ceftriaxone, up to 12 to 59% during the therapy.[21,22] In a study conducted by Scholz et al, it was observed that pseudolithiasis was commonly observed sonographically with ceftriaxone.[23] This is a wellknown occurrence with this drug and is believed to relate mainly to the precipitation of the calcium salt of ceftriaxone after secretion into bile. However, there is no such incidence observed with cefotaxime. Similarly, in a study conducted by Schaad et al, it was observed that 46% patients develop biliary pseudolithiasis. In addition, among that one case developed urolithiasis with renal colic and obstructive ureteropyelectasia.[24] This finding was later reconfirmed by many studies, where they observed formation of pseudolithiasis or sludge formation.[25,26] In fact, this cholelithiasis can turn into cholecystitis and further complication. There are three different mechanisms by which a patient receiving ceftriaxone can develop acute cholecystitis: ceftriaxone-associated sludge can trigger existing gallstones to become symptomatic, ceftriaxone pseudolithiasis can transform into ceftriaxone gallstones, or the patient can become symptomatic from preexisting cholecystolithiasis unrelated to ceftriaxone therapy. Ceftriaxone is associated with thrombocytopenia with a definite or probable causal relationship.[27] In pediatric patients, immune hemolytic anemia and biliary pseudolithiasis are major serious adverse effects found with ceftriaxone that can limit its use.[28] Ceftriaxone is associated with more incidence of Clostridium difficile infections as it is excreted in bile and have more potential to disturb gut microbiota as compared with cefotaxime, which has renal elimination. The sudden decline in incidence of C. difficile infections was noted in Leipzig University Hospital, Germany with declined and increased uses of ceftriaxone and cefotaxime, respectively.[29]
Incidence of HL-CASE Enterobacteriaceae was stabilized in France when use of ceftriaxone was replaced with cefotaxime, which was hypothesized to be associated with higher biliary elimination of ceftriaxone.[30]
CONCLUSIONS
Cefotaxime can be preferred over ceftriaxone due to its pharmacokinetic, in vitro activity, and better safety profile for empirical treatment of various infections. The present in vitro comparative study findings provide further support and lead to the conclusion that cefotaxime exhibits a superior sensitivity profile compared with ceftriaxone. This superiority is observed in terms of various parameters such as MIC, ZoI, and the sensitivity pattern of isolated organisms. The study specifically focuses on patients with UTI, LRTI, skin and soft tissue infection, sepsis, genital infections, and intraabdominal infections.
Conflict of interest
The study samples were collected and analyzed at four independent laboratories in India for which the actual payment for test charges was paid to them.
Funding
A. A. G. and D. B. P. reported financial support for this study was provided by Alkem Laboratories, Mumbai.
References
- Cefotaxime. A review of its antibacterial activity, pharmacological properties and therapeutic use. Drugs. 1983;25:223-289.
- [CrossRef] [PubMed] [Google Scholar]
- Cefotaxime: a review of in vitro antimicrobial properties and spectrum of activity. Rev Infect Dis. 1982;4:S300-S315.
- [CrossRef] [PubMed] [Google Scholar]
- Summary of Product Characteristics. Cefotaxime 1g powder for solution for injection vials Bowmed Ibisqus Limited, UK. 2021. Accessed March 22 2023 at https://www.medicines.org.uk/emc/product/8750/smpc
- [Google Scholar]
- National Treatment Guidelines for Antimicrobial Use in Infectious Diseases Version 1.0 (2016) Accessed March 22 2023 at: https://www.ncdc.mohfw.gov.in/showfile.php?lid=649
- [Google Scholar]
- Antimicrobial activity of ceftriaxone compared with cefotaxime in the presence of serum albumin. In: Can J Infect Dis. Vol 6. 1995. p. :21-27.
- [CrossRef] [PubMed] [Google Scholar]
- In vitro assay to determine the MIC90 of ß-lactam and ß-lactam-ß-lactamase inhibitor against community acquired respiratory pathogens. In: Int J Sci Stud. Vol 4. 2017. p. :93-96.
- [Google Scholar]
- District Laboratory Practice in Tropical Countries In: Part 2 (2nd ed). South Africa: Cambridge University Press; 2006. p. :1-434.
- [CrossRef] [Google Scholar]
- Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol. 1966;45:493-496.
- [CrossRef] [PubMed] [Google Scholar]
- M100-Performance Standards for Antimicrobial Susceptibility Testing. (32nd edition). Accessed March 22, 2023 at: https://clsi.org/about/press-releases/clsi-publishes-m100-performancestandards-for-antimicrobial-susceptibility-testing-32nd-edition/
- [Google Scholar]
- [Susceptibility of clinical isolates to cefotaxime] In: Jpn J Antibiot. Vol 36. 1983. p. :277-289.
- [Google Scholar]
- [Susceptibility of clinical isolates to cefotaxime. Comparison to new antibiotics latamoxef, cefoperazone, and ceftizoxime] Jpn J Antibiot. 1984;37:267-278.
- [Google Scholar]
- [The antibacterial activity of new cephem antibiotics against clinical isolates. A comparison of the antibacterial activity of cefotaxime with 6 other antibiotics] Jpn J Antibiot. 1983;36(02):260-276.
- [Google Scholar]
- Sensitivity pattern of cefotaxime against common uropathogens in vitro in Dhaka, Bangladesh. Drugs Ther Perspect. 2019;35:145-149.
- [CrossRef] [Google Scholar]
- Ceftriaxone. A review of its antibacterial activity pharmacological properties and therapeutic use. Drugs. 1984;27:469-527.
- [CrossRef] [PubMed] [Google Scholar]
- Community-and hospital-acquired infections in surgical patients at a Tertiary Referral Hospital in Rwanda. World J Surg. 2020;44:3290-3298.
- [CrossRef] [PubMed] [Google Scholar]
- [MICs and MBCs of cefotaxime, desacetylcefotaxime and ceftriaxone against four principal bacteria causing meningitis] Jpn J Antibiot. 1984;37:1701-1713.
- [Google Scholar]
- Ceftriaxone compared with cefotaxime for serious bacterial infections. J Infect Dis. 1989;160:442-447.
- [CrossRef] [PubMed] [Google Scholar]
- Cefotaxime twice daily versus ceftriaxone once daily. A randomized controlled study in patients with serious infections. Diagn Microbiol Infect Dis. 1995;22:155-157.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of cefotaxime and ceftriaxone on the bowel and vaginal flora after single-dose prophylaxis in vaginal hysterectomy. Drugs. 1988;35:163-168.
- [CrossRef] [PubMed] [Google Scholar]
- Influence of cephalosporines III generation with varying biliary excretion on fecal flora and emergence of resistant bacteria during and after cessation of therapy. Padiatr Padol. 1986;21:335-342.
- [Google Scholar]
- Prospective comparative trial of ceftriaxone vs. conventional therapy for treatment of bacterial meningitis in children. Pediatr Infect Dis. 1985;4:362-368.
- [CrossRef] [PubMed] [Google Scholar]
- Tolerance study of ceftriaxone compared with amoxicillin in patients with pneumonia. Am J Med. 1984;77:54-58.
- [Google Scholar]
- Prospective comparison of ceftriaxone and cefotaxime for the short-term treatment of bacterial meningitis in children. Chemotherapy. 1998;44:142-147.
- [CrossRef] [PubMed] [Google Scholar]
- A comparison of ceftriaxone and cefuroxime for the treatment of bacterial meningitis in children. N Engl J Med. 1990;322:141-147.
- [CrossRef] [PubMed] [Google Scholar]
- Ceftriaxone-associated biliary pseudolithiasis in children. J Clin Ultrasound. 2006;34:217-222.
- [CrossRef] [PubMed] [Google Scholar]
- Ceftriaxone-associated nephrolithiasis and gallstone in adults. Drug Healthc Patient Saf. 2018;10:103-108.
- [CrossRef] [PubMed] [Google Scholar]
- A systematic evaluation of laboratory testing for drug-induced immune thrombocytopenia. J Thromb Haemost. 2013;11:169-176.
- [CrossRef] [PubMed] [Google Scholar]
- Safety of ceftriaxone in paediatrics: a systematic review. Arch Dis Child. 2020;105:981-985.
- [CrossRef] [PubMed] [Google Scholar]
- Switching from ceftriaxone to cefotaxime significantly contributes to reducing the burden of Clostridioides difficile infections. Open Forum Infect Dis. 2020;7:ofaa312.
- [CrossRef] [PubMed] [Google Scholar]
- Fighting the spread of AmpC-hyperproducing Enterobacteriaceae: beneficial effect of replacing ceftriaxone with cefotaxime. J Antimicrob Chemother. 2014;69:786-789.
- [CrossRef] [PubMed] [Google Scholar]





