Translate this page into:
C4d immunohistochemistry in membranous nephropathy
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background:
Membranous nephropathy (MN) is the most common cause of nephropathy in adults. The diagnosis is based on characteristic light microscopic, electron microscope and immunofluorescence (IF) findings. In early MN, the light microscopic findings may be difficult to differentiate from minimal chain disease. In the absence of fresh frozen tissue for IF, immunohistochemistry with C4d aids in the diagnosis.
Materials and Methods:
A total 48 cases of MN diagnosed on renal biopsy were analyzed. The formalin fixed paraffin embedded tissues were stained with routine hematoxylin and eosin stains along with periodic acid-Schiff and silver methenamine stains to highlight the basement membrane. Fresh frozen tissues were available for IF in 40 cases. Immunostaining with C4d was done on paraffin-embedded sections by polymer-Horse Radish Peroxidase (HRP) technique using polyclonal antiserum to C4d (Biogenex, India).
Results:
There were 25 cases of idiopathic MN, 17 cases of Class V lupus nephritis and 2 cases were secondary to hepatitis C infection with cirrhosis. The glomerular basement membrane (GBM) was diffusely thickened with formation of spikes in 28 cases. In 11 cases the capillary loops were rigid but spikes were not seen and in 9 cases there was no apparent thickening of the basement membrane. All the cases showed diffuse positivity for C4d along the GBM.
Conclusion:
C4d is a reliable method to establish the diagnosis of MN and also a sensitive marker of complement activation reflecting the pathogenesis of MN.
Keywords
C4d
complement activation
immunohistochemistry
membranous nephropathy
INTRODUCTION
The differential diagnosis of membrane thickening in renal biopsies includes primary and secondary membranous nephropathy (MN), diabetic nephropathy and amyloidosis. Of these, MN is the most common cause of nephrotic syndrome in adults accounting for 20-30% cases. The diagnosis is based on clinical features and characteristic light microscopic findings of diffuse thickening of glomerular capillary wall. However, in early membranous cases, the membrane thickening and spikes may not be obvious. In such situation immunofluorescence (IF) examination is necessary to differentiate from minimal change disease (MCD) where in MN shows granular staining with immunoglobulin G (IgG) and or C3. In the absence of availability of fresh tissues or absence of glomeruli in the frozen tissue, the diagnosis of MN cannot be confirmed. Immunohistochemistry (IHC) with C4d has been reported as a novel marker for diagnosis of MN in various published studies.[123] In addition to its diagnostic utility, it also adds insight to pathogenesis of MN. MN is characterized by deposition of immune complexes in sub epithelial region of the glomerular capillary wall. The formation of these in situ immune complexes results in activation of complement which generates formation of C4d. It binds to the epithelial and endothelial cell surfaces and can be used as a marker of complement activation in MN. In this study, we try to evaluate the utility of C4d IHC in diagnosis of MN.
MATERIALS AND METHODS
Consecutive of 48 cases of MN diagnosed on renal biopsy from August 2012 to April 2013 were included in the study. Age, gender, clinical presentation and laboratory findings were documented in every case. The renal biopsies were stained and studied with hematoxylin and eosin, periodic acid-Schiff (PAS), Masson trichrome and silver methanamine-PAS stains. Fresh frozen tissues were available for IF in 40 cases. IF study was performed with anti-IgG, IgM, IgA, C3, C1q, kappa, and lambda antibodies. All the cases were immunostained with C4d antibodies (Monoclonal, Biogenex). MCD, diabetic nephropathy and amyloidosis were chosen as a control with the addition of 4 cases each of Class III and Class IV lupus nephritis (LN) [Figure 2] since C4d has earlier been reported positive in proliferative LN.[4]
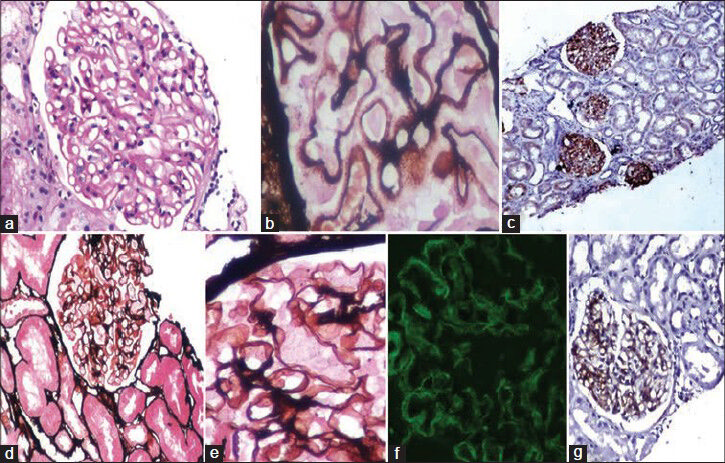
- Classic case of membranous nephropathy with (a) thickened basement membrane, (b) showing spikes on silver methanamine-periodic acid-Schiff (SM-PAS) stain and (c) granular positivity for C4d along the basement membrane. Horseradish peroxidase polymer; C4d. Another biopsy of a 36/M with 24 h proteinuria of 3.5 g. The glomerulus on SM-PAS stain, (d and e) showing almost normal glomerulus without apparent thickening of the basement membrane, (f) immunofluorescence showed granular positivity for immunoglobulin G, (g) however C4d showed positivity along the glomerular basement membrane
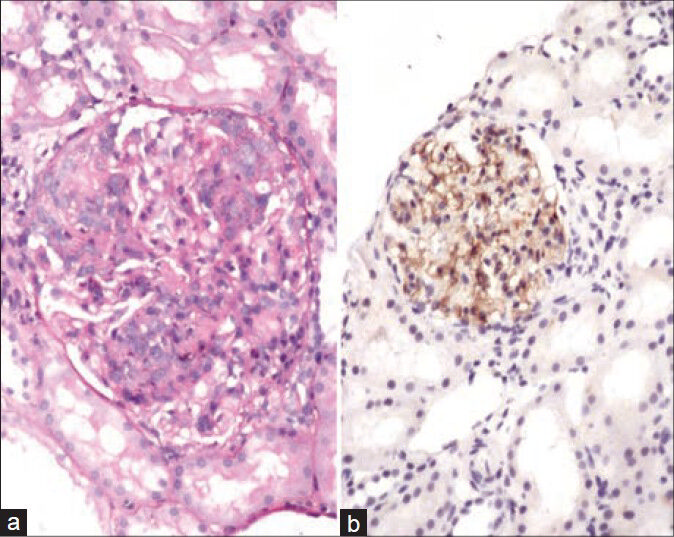
- (a) A case of class IV lupus nephritis with diffuse proliferation and neutrophils, (b) C4d positivity along the glomerular basement membrane
RESULTS
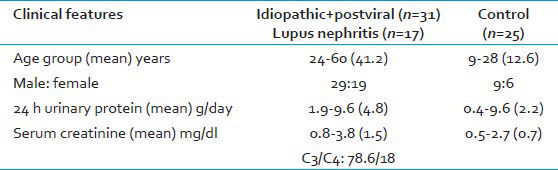
The study group of MN was formed by 25 cases of primary MN, 2 secondary MN (hepatitis C with cirrhosis) and 17 of Class V LN. The patients were in the age range of 14-60 years with male:Female ratio of 1.53:1. The clinical presentations included edema (41), proteinuria (43), hematuria (8), oliguria (4), renal dysfunction (9) and nephrotic syndrome (33). Six of these were hypertensive, and none were diabetic. The clinical and investigative details of the patients and controls are provided in Table 1. Separate details of patients with LN are given in Tables 2 and 3.

Light microscopic evaluation showed characteristic membrane thickening with spikes in 28 cases [Figure 1] whereas 11 biopsies had only rigidity of capillary loops without spikes or holes on silver stain. In 9 biopsies, the glomeruli had near normal appearance without apparent thickening or spikes. IF examination showed positivity for IgG and C3 where ever available. Of the classic MN cases, fresh frozen tissue for IF was not available in 8 and in 2 there were no glomeruli in the tissue submitted for IF. However, IHC done on paraffin fixed tissues showed granular positivity of C4d along the glomerular basement membrane (GBM) in 100% cases. All the control groups including 15 cases of MCD, 3 cases of diabetic nephropathy and 2 cases of amyloidosis were negative for C4d.
DISCUSSION
C4d is a degradation product of complement factor C4, which is activated during the classical, as well as lectin pathway of complement cascade. The classical pathway of complement is activated by conformational changes in Ig molecules, after binding to specific antigens. This is followed by cleavage of C4 domains into C4a and C4b. C4b is then cleaved by C3bINA in the presence of C4b binding proteins into C4c and C4d. C4d covalently binds to cell surfaces via exposed thiolester group.[4] Antibodies dissociate naturally because of relatively weak hydrostatic and van der Waals forces between antigens and antibodies, whereas covalent bond of C4d has a much longer half-life. Therefore, detection of C4d is regarded as an indirect sign, a “footprint” of complement activation.[5]
The use of IHC to detect the C4d complement degradation product in kidney disease has sparked considerable clinical interest recently. C4d positivity in peritubular capillaries is a diagnostic marker for antibody mediated rejection as incorporated by Banff 2007.[6] The literature on its use in glomerulonephritis, is however limited. Recently the role of C4d was highlighted in diagnosis and pathogenesis of IgA nephropathy, MPGN and MN.[1] The other studies on C4d in MN has have shown near 100% positivity similar to what was observed by us in this study.[123]
The original description of the pathogenesis of MN includes role of 4 auto antigens like M type phospholipase A2 receptor, aldose reductase, manganese superoxide dismutase, membrane metalloendopeptidase. Complement activation with resultant C5b-9 complex is a known fact in MN.[7] C4d is only an extension of our understanding of complement activation in immune complex glomerulonephritis. Its presence can also be extrapolated in the form of IHC for diagnostic purpose, which is an added advantage.
Minimal change disease did not show positivity in any of the cases. It is well known that MCD is not an immune complex disorder and occurs due to podocyte effacement. Negativity for C4d rules out complement activation in MCD. Negativity of C4d in diabetic nephropathy and amyloidosis also stresses the role of C4d in diagnosis of MN since all these diseases can have similar clinical background however requires different management.
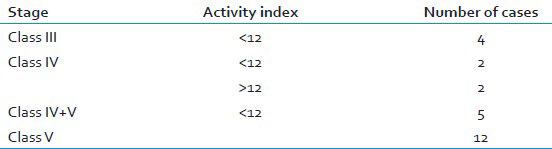
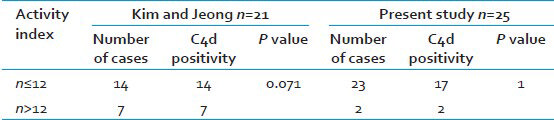
Glomerular C4d is similarly a marker of complement pathway activation in LN. C4d was detected by Kim and Jeong in 21 cases of LN however did not correlate with disease activity.[4] In our study, all the cases of Class V LN and 2 of Class IV were positive for C4d. Hence To conclude C4d as a marker of Class V disease or disease activity is difficult in view of small sample size. However, C4d can be of help for diagnosis of Class V disease in correlation with clinical features since follow-up LN biopsies usually are not submitted for IF. The comparison of the results of our study and Kim and Jeong has been given in Table 4.
The interpretation of IHC would be incomplete without stressing on the site of positivity of C4d. In our study C4d deposits were located in the GBM with a granular distribution indicating the site of complement activation with formation of immune complex deposits. IgA nephropathy shows mesangial positivity for C4d and correlated with poor prognosis. Espinosa et al. also found a higher rate of renal survival after 10 years in C4d negative cases as compared to C4d positive cases.[8] In a recent study of 283 cases of IgA nephropathy by the same author in 2014, the survival of patients at 20 years was 28% in C4d-positive patients as compared to 85% in C4d-negative patients.[9]
Various studies have compared the two techniques of IF and IHC for their sensitivity and specificity in diagnosis of renal diseases.[1011] It has been observed that the results of IF and IHC are comparable with respect to pattern and intensity. Suzuki et al. has stressed the advantage of C4d IHC in glomerular diseases as compared to IF.[10] IHC has a particular advantage of being carried out on paraffin embedded tissue whereas IF needs fresh frozen tissue.
CONCLUSION
C4d is a reliable method to establish the diagnosis of MN and a useful marker to differentiate early MN from MCD. It may obviate the need of rebiopsy if tissue is not available for IF study. C4d immunostaining is also a sensitive marker of complement activation which reflects the pathogenesis of MN.
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- Glomerular deposition of C4 cleavage fragment (C4d) and C4-binding protein in idiopathic membranous glomerulonephritis. Nephron. 1989;51:17-9.
- [Google Scholar]
- C4d immunohistochemical staining is a sensitive method to confirm immunoreactant deposition in formalin-fixed paraffin-embedded tissue in membranous glomerulonephritis. Histol Histopathol. 2011;26:1391-7.
- [Google Scholar]
- Glomerular C4d deposition indicates in situ classic complement pathway activation, but is not a marker for lupus nephritis activity. Yonsei Med J. 2003;44:75-80.
- [Google Scholar]
- Banff 07 classification of renal allograft pathology: Updates and future directions. Am J Transplant. 2008;8:753-60.
- [Google Scholar]
- An overview of molecular mechanism of nephrotic syndrome. Int J Nephrol 2012 2012:937623.
- [Google Scholar]
- Mesangial C4d deposition: A new prognostic factor in IgA nephropathy. Nephrol Dial Transplant. 2009;24:886-91.
- [Google Scholar]
- Association of C4d deposition with clinical outcomes in IgA nephropathy. Clin J Am Soc Nephrol. 2014;9:897-904.
- [Google Scholar]
- C4d immunohistochemistry in glomerulonephritis with different antibodies. Clin Exp Nephrol. 2007;11:287-91.
- [Google Scholar]
- Immunoperoxidase versus immunofluorescence in the assessment of human renal biopsies. Am J Kidney Dis. 2005;45:674-83.
- [Google Scholar]






