Translate this page into:
Evaluation of different phenotypic tests for detection of metallo-β-lactamases in imipenem-resistant Pseudomonas aeruginosa
-
Received: ,
Accepted: ,
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
PURPOSE:
Pseudomonas aeruginosa causes a wide spectrum of infections including bacteremia, pneumonia, urinary tract infection, etc., Metallo-beta-lactamase (MBL) producing P. aeruginosa is an emerging threat and cause of concern as they have emerged as one of the most feared resistance mechanisms. This study was designed to know the prevalence of MBL production in P. aeruginosa and to evaluate the four phenotypic tests for detection of MBL production in imipenem-resistant clinical isolates of P. aeruginosa.
METHODS:
Totally, 800 isolates of P. aeruginosa isolated from various clinical samples were evaluated for carbapenem resistance and MBL production. All imipenem-resistant strains were tested for carabapenemase production by modified Hodge test. Screening for MBL production was done by double-disc synergy test and combined disc test (CDT). Confirmation of MBL production was done by the E-test (Ab BioDisk, Solna, Sweden).
RESULTS:
Out of the 800 isolates of P. aeruginosa, 250 isolates were found resistant to imipenem. Based on the results of E-test, 147 (18.37%) isolates of P. aeruginosa were positive for MBL production. The CDT has the highest sensitivity and specificity for the detection of MBL production as compared to other tests.
CONCLUSION:
The results of this study are indicative that MBL production is an important mechanism of carbapenem resistance among P. aeruginosa. Use of simple screening test like CDT will be crucial step toward large-scale monitoring of these emerging resistant determinants. Phenotypic test for MBL production has to be standardized, and all the isolates should be routinely screened for MBL production.
Keywords
Combined disc test
double-disc synergy test
E-test
modified Hodge test
Introduction
Pseudomonas aeruginosa is a leading cause of nosocomial infections worldwide. The metabolic ingenuity of P. aeruginosa contributes to its broad ecological adaptability, ubiquitous distribution, ability to acquire and disseminate resistance vertically and horizontally in the hospital environment, and tendency to remain viable on both animate and inanimate objects around the patient including antiseptic solutions.[1]
Carbapenems were introduced in the 1980s and are used as last choice in treating serious infections caused by multidrug-resistant Gram-negative bacilli. These antibiotics are stable to β-lactamase including extended spectrum beta-lactamases, and AmpC produced by Gram-negative bacilli.[2345] Resistance to these antibiotics started emerging from the 1990s.[2] Resistance to carbapenem in P. aeruginosa is due to decreased outer membrane permeability, increased efflux system, alteration of penicillin-binding protein and carbapenem-hydrolyzing enzymes carbapenemases. These carbapenemases are Class B metallo-beta-lactamase (MBL) or Class D oxacillinase (OXA 23 to OXA 25) or Class A clavulanic acid inhibiting enzyme.[678]
MBL producing P. aeruginosa is an emerging threat and a cause of concern as they have emerged as one of the most feared resistance mechanisms. MBL belongs to Ambler Class B owing to their capacity to hydrolyze all β-lactams including penicillins, cephalosporins and carbapenems with the exception of aztreonam.[8] They differ from other carbapenemases in having broad substrate profile, potential for horizontal transfer, and lack of inhibition by serine β-lactamase inhibitors such as clavulanic acid, sulbactam, and tazobactam.[9] They require divalent metal ion for enzymatic activity.[10] MBL genes are normally encoded in class 1 integrons along with other resistance determinants, such as the aminoglycoside-modifying enzymes. The integrons are frequently located in plasmids or transposons, which contributes to the overall spread of this resistance mechanism. Different types of MBL are known in P. aeruginosa, including IMP, VIM, German imipenemase, Sao Paulo MBL, Seoul imipenemase, New Delhi MBL, and Adelaide imipenemase 1. The most common and widespread acquired MBL are those of the IMP and VIM types, which exhibit a worldwide distribution.[11]
MBL production in P. aeruginosa can be detected by molecular methods and phenotypic methods. In molecular methods, polymerase chain reaction (PCR), DNA probes, and cloning and sequencing can be done to detected MBL-positive genes. These methods are highly accurate and reliable, but they are available only in reference laboratories.
The phenotypic methods of MBL production are based on the ability of metal chelators such as EDTA and thiol-based compounds to inhibit the activity of MBL. The common methods are modified Hodge test (MHT), double-disc synergy test (DDST), and combined disc diffusion test using imipenem and EDTA, and MBL E-test (Ab BioDisk, Solna, Sweden).
This study was designed to know the prevalence of MBL production in P. aeruginosa and to evaluate the four phenotypic tests for the detection of MBL production in imipenem-resistant clinical isolates of P. aeruginosa.
Methods
This study was conducted from January 2012 to December 2012. Totally, 800 isolates of P. aeruginosa isolated from various clinical samples were evaluated for carbapenem resistance and MBL production. The isolates were identified by standard laboratory techniques.[12] All these isolates were screened for imipenem resistant by Kirby-Bauer disc diffusion method as per Clinical and Laboratory Standard Institute guidelines.[13] All imipenem-resistance strains were tested for carabapenemase production by MHT.[14] Screening for MBL production was done by DDST[15] and combined disc test (CDT).[16] Confirmation of MBL production was done by the E-test (Ab BioDisk, Solna, Sweden).
Modified Hodge test
A lawn culture of 1:10 dilution of 0.5 McFarland's standard Escherichia coli ATCC 25922 was done on a Muller-Hinton (MH) agar. A 10 μg imipenem disc (Hi-media) was placed in the center of the plate. Imipenem-resistant P. aeruginosa (test isolates) were streaked from the edge of the disc to the periphery of the plate in four different directions. After overnight incubation, the plates were observed for the presence of a “clover-leaf” shaped zone of inhibition which was interpreted as MHT positive.
The double-disc synergy test
An overnight culture of the test isolate (0.5 McFarland opacity standard) was inoculated on a MH agar. After drying, a 10 μg imipenem disc and a sterile blank disc (Hi-media) were placed 10 mm apart from edge to edge. Another imipenem disc was placed far as control. A volume of 10 μl of 0.5 M EDTA (Sigma, USA) solution was applied to the blank disc. After overnight incubation, the zone of inhibition around imipenem disc expands toward EDTA disc, compared to the other imipenem disc, placed on the far side was interpreted as a positive result.
Combined disc diffusion method (combined disc test)
A lawn culture of test isolate (0.5 McFarland opacity standard) was done on MH agar. Two 10 μg imipenem discs were placed on inoculated plates. To one of the imipenem discs, 10 μl of 0.5 M EDTA solution was added. After overnight incubation, if the zone of inhibition of imipenem + EDTA discs compared to imipenem alone is >7 mm, the test was considered as positive.
E-test
An E-test MBL strip contains a double-sided seven-dilution range of imipenem (4–256 μg/ml) and imipenem (1–64 μg/ml) in combination with a fixed concentration of EDTA. A lawn culture of test isolate of 0.5 McFarland opacity standard was done on MH agar. E-test strip was inoculated on the surface of agar. After overnight incubation, the plates were observed for imipenem and imipenem – EDTA minimum inhibitory concentration (MIC) values, where the respective inhibition ellipses intersect the strip. The test was considered MBL positive when the MIC ratio of imipenem/imipenem plus EDTA was more than eight. The presence of a phantom zone or a deformation of the imipenem ellipse was also considered as positive.
Results
Out of the 800 isolates of P. aeruginosa collected in this study, 250 isolates were found resistant to imipenem. All the imipenem resistant isolates were tested for MBL production by three different methods. Based on the results of E-test, 147 (18.37%) isolates of P. aeruginosa were positive for MBL production. Table 1 gives the comparison of three different tests for MBL production against the confirmatory E-test. None of the test methods showed complete correlation when compared to the confirmatory E-test. The CDT has the highest sensitivity and specificity for detection of MBL production as compared to other tests [Figure 1].

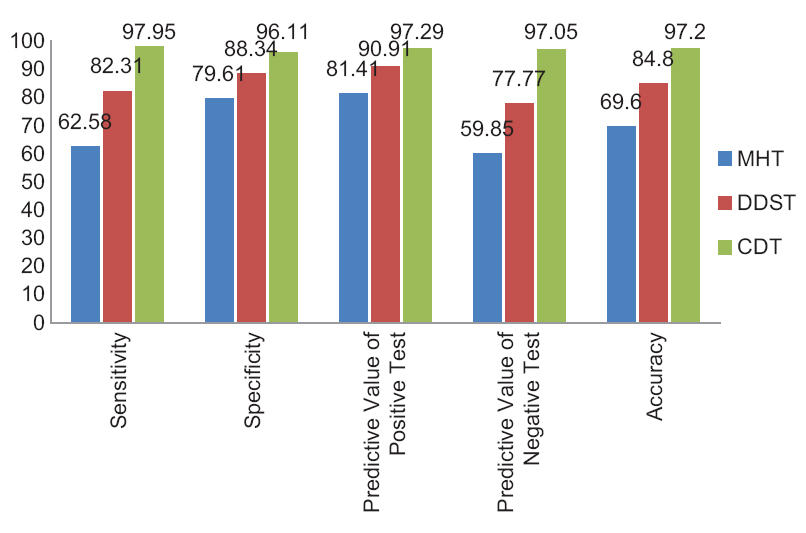
- Performance parameter of different phenotypic tests for metallo-beta-lactamase production. Note: MHT = Modified Hodge test, DDST = Double-disc synergy test, CST = Combined disc test
Discussion
P. aeruginosa is an epitome of opportunistic nosocomial pathogen which causes a wide spectrum of infections including bacteremia, pneumonia, urinary tract infection, etc. Multiple factors contribute to make P. aeruginosa as nosocomial pathogen such as injudicious administration of broad-spectrum antibiotics, instrumentation, and intrinsic resistance of microorganism to numerous antimicrobial agents.[17]
Several phenotypic methods are available for the detection of MBL producing bacteria. All these methods are based on the ability of metal chelators such as EDTA and thiol-based compounds to inhibit the activity of MBLs. In this study, we compared four phenotypic methods for the detection of MBL production in all imipenem-resistant P. aeruginosa.
The MBL producing P. aeruginosa was first reported from Japan in 1988[18] and since then has been reported from various parts of the world. In India, MBL was first reported in 2002 by Navaneeth et al.[19] Since that the incidence of MBL production in P. aeruginosa has been reported from various parts of India.
In the present study, 250 isolates of P. aeruginosa resistant to imipenem were subjected for MBL production by four different phenotypic methods. Out of these 250 isolates, 147 (18.37%) isolates of P. aeruginosa were positive for MBL production by E-test. Our results are consistent with Murugan et al.[20] and Varaiya et al.[21] However, few studies reported higher incidence of MBL producers. In the present study, 58.8% of imipenem-resistant strains were positive for MBL production. It shows that some other mechanisms of imipenem resistance does occur other than MBL production such as loss of the porin OprD, activity of chromosomal β-lactamase (AmpC), or increased expression of efflux pump system.
MBL E-test is a sensitive method for detection of MBL in P. aeruginosa. The E-test, based on a combination of a β-lactam substrate and a β-lactam/MBL inhibitor, is specifically designed to detect as many clinically relevant MBL as possible. The E-test has the ability to detect MBLs, both chromosomally and plasmid-mediated in aerobic and anaerobic bacteria.[22] E-test also determines the MIC. In our study, all E-test positive MBL strains were having MIC of >16 μg/ml for imipenem, and phantom zone was noted in 26 isolates. Our study is comparable to that of Behera et al.[23] The test is simple to perform and easy to interpret. However, there are conflicting reports regarding the performance of MBL E-test in literature. Khosravi et al.[24] reported 100% accuracy of MBL E-test with PCR for the detection of MBL production. The discrepancy in finding may be due to difference in population structure of MBL gene between different geographical area.[24] The MBL E-test has been reported to be insensitive to detect carbapenem sensitive MBL carrying organism.[25] In our study, we screened only carbapenem-resistant isolates which may account for very high sensitivity of the test.
MHT is used for screening carbapenemases activity and indicates enzyme production. It cannot distinguish MBL carbapenemases from non-MBL carbapenemases such as oxacillinases production. In our study, it showed 62.58% and 79.6% sensitivity and specificity, respectively. It was comparable to that of Franco et al.[1] This test can be used as a screening test for MBL production, but it is difficult to interpret. Imipenem-resistant strains were found to be carbapenem hydrolysis negative by MHT. The reason for imipenem resistance is mechanism other than carbapenem hydrolysis such as decreased membrane permeability.
In our study, out of 147 MBL-positive isolates, 121 were detected by DDST. Double-disc synergy test seems to be a better method for MBL detection then MHT. Similar results have been observed by Franco et al.,[1] DDST can be used a screening test for MBL production. The only disadvantage of DDST was the subjective interpretation of results.
The CDT using imipenem + EDTA was found to be superior to DDST. We also found CDT as to be very sensitive for the detection of MBL in P. aeruginosa. With the imipenem and EDTA CDT with a cutoff >7 mm, the positive and negative results were more clearly discriminated. Our finding is in accordance with other published study of Franklin et al.[25] and Manoharan et al.,[26] CDT for MBL production is simple to perform and materials used are cost-effective, nontoxic, and easily available, which makes it highly pertinent as screening test in routine clinical laboratory.
In the evaluation of three selected MBL phenotypic assays (MHT, DDST, and CDT), there is difference in sensitivity and specificity. The MHT has least sensitivity and specificity. The CDT has 97.95% sensitivity and 96.11% specificity when compare with E-Test. Our results are comparable with that of Qu et al.,[27] who also demonstrated that the CDT is the best method for screening for MBL production in P. aeruginosa. However, Ranjan et al.,[28] Lee et al.,[15] and Pitout et al.[29] demonstrated that DDST was more specific in detecting MBLs in comparison to the CDT. This discrepancy in findings may be due to differences in population structure of MBL genes between different geographical areas. False-positive MBL producers were detected by all phenotypic assays. These false-positive cases might actually be producing an unknown and weaker β-lactamases, which should be further investigation.
The incidence of MBL's has been steadily increasing, and it is disseminating widely. The detection of MBL is of utmost importance in deciding the most appropriate therapeutic requirement. The correlation between carriage of MBL gene and carbapenem resistance is imperfect as either MBL gene are not always expressed or substantive resistance may require uptake of carbapenem as well the presence of MBL. As MBL is also reported in carbapenem sensitive isolates ideally, all isolates should be screened for MBL production. We screened only carbapenem-resistant isolate as it is most often preferred, but it is suboptimal however screening all isolates will increase the workload with a lower yield. We recommended that all isolates of P. aeruginosa resistant to imipenem, ceftazidime, and piperacillin + tazobactam should be routinely screened for MBL production by CDT since this test is simple to perform and interpret. It is performed as routine antimicrobial susceptibility method as it can be easily introduced into the workflow of a clinical laboratory. It is less expensive than the MBL E-test.
Conclusion
MBL production is an important mechanism of carbapenem resistance among P. aeruginosa. Although PCR gives specific and accurate results, its use is limited to only a few laboratories because of its high cost and different types of MBLs which are present worldwide. The E-test is very sensitive for the detection of MBL in P. aeruginosa. It may not be practically possible for all laboratories to perform the E-test due to cost constraints and availability. Use of simple screening test like CDT will be crucial step toward large-scale monitoring of these emerging resistant determinants.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- The determinants of the antibiotic resistance process. Infect Drug Resist. 2009;2:1-11.
- [Google Scholar]
- Incidence of carbapenem resistant nonfermenting Gram negative bacilli from patients with respiratory infections in the Intensive Care Units. Indian J Med Microbiol. 2005;23:189-91.
- [Google Scholar]
- Sensitivities to four carbapenems of bacteria isolated from patients with refractory complicated urinary tract infections and the detection of carbapenemase-producing Pseudomonas aeruginosa. J Antimicrob Chemother. 1996;38:322-4.
- [Google Scholar]
- Carbapenem activities against Pseudomonas aeruginosa: Respective contributions of OprD and efflux systems. Antimicrob Agents Chemother. 1999;43:424-7.
- [Google Scholar]
- Interplay of impermeability and chromosomal beta-lactamase activity in imipenem-resistant Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1992;36:2046-8.
- [Google Scholar]
- Metallo-beta-lactamases: The quiet before the storm? Clin Microbiol Rev. 2005;18:306-25.
- [Google Scholar]
- A functional classification scheme for beta-lactamases and its correlation with molecular structure. Antimicrob Agents Chemother. 1995;39:1211-33.
- [Google Scholar]
- Metallo beta lactamases in Pseudomonas aeruginosa and Acinetobacter species. Expert Opin Investig Drugs. 2008;17:131-43.
- [Google Scholar]
- Tests for the identification of bacteria. In: Colle JG, Fraser AG, Marimon BP, Simmons A, eds. Mackei and McCartney Practical Medical Microbiology (14th ed). Edinburg: Churchill Livingstone; 1996. p. :131-50.
- [Google Scholar]
- Performance Standards for Antimicrobial Susceptibility Testing. Twentieth Informational Supplement. In: CLSI Document M100-S20. Wayne, PA: CLSI; 2010.
- [Google Scholar]
- Modified Hodge and EDTA disk synergy test to screen metallo-beta-lactamases producing strains of Pseudomonas spp and Acinetobacter spp. Clin Microbiol Infect. 2001;7:88-91.
- [Google Scholar]
- Evaluation of the Hodge test and the imipenem-EDTA double-disk synergy test for differentiating metallo-beta-lactamase-producing isolates of Pseudomonas spp. and Acinetobacter spp. J Clin Microbiol. 2003;41:4623-9.
- [Google Scholar]
- Imipenem-EDTA disk method for differentiation of metallo-beta-lactamase-producing clinical isolates of Pseudomonas spp. and Acinetobacter spp. J Clin Microbiol. 2002;40:3798-801.
- [Google Scholar]
- Mechanisms of beta-lactam resistance amongst Pseudomonas aeruginosa isolated in an Italian survey. J Antimicrob Chemother. 1998;42:697-702.
- [Google Scholar]
- Transferable imipenem resistance in Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1991;35:147-51.
- [Google Scholar]
- A preliminary study on metallo-beta-lactamase producing Pseudomonas aeruginosa in hospitalized patients. Indian J Med Res. 2002;116:264-7.
- [Google Scholar]
- Prevalence and antimicrobial susceptibility pattern of metallo beta lactamase producing Pseudomonas aeruginosa in diabetic foot infection. Int J Microbiol Res. 2010;1:123-8.
- [Google Scholar]
- Incidence of metallo beta lactamase producing Pseudomonas aeruginosa in ICU patients. Indian J Med Res. 2008;127:398-402.
- [Google Scholar]
- Evaluation of a new Etest for detecting metallo-beta-lactamases in routine clinical testing. J Clin Microbiol. 2002;40:2755-9.
- [Google Scholar]
- An evaluation of four different phenotypic techniques for detection of metallo-beta-lactamase producing Pseudomonas aeruginosa. Indian J Med Microbiol. 2008;26:233-7.
- [Google Scholar]
- Phenotypic detection of metallo-ß-lactamase in imipenem-resistant Pseudomonas aeruginosa. ScientificWorldJournal. 2012;2012:654939.
- [Google Scholar]
- Phenotypic detection of carbapenem-susceptible metallo-beta-lactamase-producing gram-negative bacilli in the clinical laboratory. J Clin Microbiol. 2006;44:3139-44.
- [Google Scholar]
- SARI Study Group. Detection and characterization of metallo beta lactamases producing Pseudomonas aeruginosa. Indian J Med Microbiol. 2010;28:241-4.
- [Google Scholar]
- Evaluation of phenotypic tests for detection of metallo-beta-lactamase-producing Pseudomonas aeruginosa strains in China. J Clin Microbiol. 2009;47:1136-42.
- [Google Scholar]
- Evaluation of phenotypic tests and screening markers for detection of metallo-beta lactamase in clinical isolates of Pseudomonas aeruginosa: A prospective study. Med J DY Patil Univ. 2015;8:599-605.
- [Google Scholar]
- Detection of Pseudomonas aeruginosa producing metallo-β-lactamases in a large centralized laboratory. J Clin Microbiol. 2005;43:3129-35.
- [Google Scholar]






