Translate this page into:
Impact of Red Cell and Platelet Distribution Width in Patients of Medical Intensive Care Unit
This article was originally published by Thieme Medical and Scientific Publishers Pvt. Ltd. and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Introduction
The red blood cell distribution width (RDW) is a measurement of variations in the size of red blood cells. As the width increases, the rate of mortality also increases, although the reason for it is still not known. On the other hand, platelet distribution width (PDW) is also useful in predicting morbidity and mortality in sepsis and other critically ill patients. In our study, we planned to study the impact of both RDW and PDW and evaluate their prognostic importance with outcome in patients admitted in medicine intensive care unit (MICU).
Material and Method
In these cross-sectional observational studies, 1,300 patients were included who were admitted in MICU. Critically ill patients were defined on the basis of qSOFA score greater than 2. PDW and RDW were obtained from Coulter report of complete blood count. Parameters included in Acute Physiology and Chronic Health Evaluation (APACHE) IV scores were taken and APACHE IV score was calculated. Correlation of RDW and PDW with outcomes such as length of ICU stay, use of mechanical ventilator, and discharge/death was done.
Result
The mean RDW (%) for the entire study population was 15.17 ± 3.01. The RDW (%) was significantly on the higher side in patients who succumbed to the disease as compared with the patients who were discharged. The correlation between the length of ICU stay (days) and RDW (%) was moderately positive and was significant (rho = 0.37, p ≤ 0.001). The correlation between the length of ICU stay (days) and PDW (%) was moderately positive and was significant (rho = 0.5, p ≤ 0.001).
Conclusion
RDW and PDW were found as significant indicators for period of stay in ICU, requirement for mechanical ventilation, and mortality rate in patients admitted to ICUs. As these are simple, easy to conduct, universally available tests, they can be regularly incorporated in patients admitted in ICUs.
Keywords
red cell distribution width
platelet distribution width
mortality
morbidity
critically ill
Introduction
The red blood cell distribution width (RDW) is a measurement of variations in the size of red blood cells.[1] Width of platelet distribution (PDW) is an index of platelet which tells about variation in size of platelet.[2] Normal range for PDW is 10 to 17%.[3] Erythrocytes have variation in size, which becomes tiny during aging.[4] Recent studies had reported associations of RDW and PDW in patients with community-acquired pneumonia, cardiac failure, pulmonary hypertension, and myocardial infarction in the general population.[5-9] As RDW increases, the rate of mortality also increases and the reason for it is still not known. In few studies it was suggested that an increased RDW is due to inflammatory state.[10]
There is always a change in the morphology on activation of platelet in the process of inflammation.[11,12]Erez et al proposed that there was more activation of thrombin during certain critically ill conditions due to which there was morphological alteration in platelet, and they appeared larger as they became spherical in shape and has developed pseudopodia.[13] As a result, platelets with greater number of large size pseudopodia will cause variation in PDW. Hypercoagulability is other possible mechanism for higher variation in platelet size.[14] Ahmad and Waheed showed in his study that newborn with sepsis of late onset had a massive rise in PDW.[15] Although few templates have been assessed to study the mortality and morbidity in intensive care units (ICUs) regarding RDW and PDW, they have been studied separately. In our study we have tried to study the impact of both RDW and PDW combined and to evaluate its prognostic importance with hospital mortality in patients who are critically ill; to predict outcome in patients admitted in medical intensive care units (MICUs) in terms of length of ICU stay, need for ventilator, and mortality; to compare red cell distribution width and platelet distribution width with Acute Physiology and Chronic Health Evaluation (APACHE) IV score.
Material and Methods
This cross-sectional study was accomplished in the MICU under Medicine department from October 2018 to July 2020 at rural teaching hospital at central India after clearance from the Institutional Ethical Committee with approval letter numbered DMIMS (DU)/IEC/2018–19/7555. All the consecutive/sequential patients regardless of gender, ethnicity, or reason for hospitalization who were admitted under MICU with qSOFA score ≥ 2 were taken as critically ill and were included in the study. PDW and RDW were obtained from complete blood count Coulter report; parameters needed for calculation of APACHE IV scores were taken and calculation of APACHE IV score was done using internet-based online module. Correlation of RDW and PDW with outcome such as the use of mechanical ventilator, length of ICU stays and discharge, and death was done. Patients were transferred from another (surgical) ICU. Patients readmitted to MICU during the same hospital stay were excluded from the study. Flowchart showing plan of study is shown in ►Fig. 1

- Flow chart showing plan of study.
Result
►Table 1 illustrates baseline characteristics of total 1,300 patients according to APACHE IV category. The mean age for the patient in the score group of 0 to 99 was 46.38 ± 18.14 while in 100 to 199 group was 50.87 ± 16.59 and in 200 to 286 group was 57.25 ± 17.11. In 0 to 99 group 230 patients were male and 141 patients were female while in 100 to 199 group 428 patients were male and 258 patients were female and in 200 to 286 group 167 patients were male and 76 were female. The mean RDW (%) in 0 to 99 score group was 14.28 ± 0.16 while in 100 to 99 score group it was 14.94 ± 3.87 and in 200 to 286 group it was 17.18 ± 0.82. The mean PDW (%) in 0 to 99 score group was 9.29 ± 0.23 while in 100 to 99 score group it was 11.23 ± 1.39 and in 200 to 286 group it was 15.64 ± 0.59.
| Parameters | APACHE IV score category | p-Value | ||
|---|---|---|---|---|
| 0–99 (n = 371) | 100–199 (n = 686) | 200–286 (n = 243) | ||
| Age (Years)*** | 46.38 ± 18.14 | 50.87 ± 16.59 | 57.25 ± 17.11 | < 0.0011 |
| Age*** | ||||
| < 40 y | 146 (39.4%) | 168 (24.5%) | 40 (16.5%) | < 0.0012 |
| 40–60 y | 124 (33.4%) | 306 (44.6%) | 95 (39.1%) | |
| > 60 y | 101 (27.2%) | 212 (30.9%) | 108 (44.4%) | |
| Gender | ||||
| Male | 230 (62.0%) | 428 (62.4%) | 167 (68.7%) | 0.1662 |
| Female | 141 (38.0%) | 258 (37.6%) | 76 (31.3%) | |
| Outcome*** | ||||
| Discharge | 368 (99.2%) | 666 (97.1%) | 11 (4.5%) | < 0.0012 |
| Death | 3 (0.8%) | 20 (2.9%) | 232 (95.5%) | |
| APACHE IV score*** | 76.16 ± 8.47 | 126.35 ± 20.61 | 250.35 ± 18.55 | < 0.0011 |
| RDW (%)*** | 14.28 ± 0.16 | 14.94 ± 3.87 | 17.18 ± 0.82 | < 0.0011 |
| PDW (%)*** | 9.29 ± 0.23 | 11.23 ± 1.39 | 15.64 ± 0.59 | < 0.0011 |
| Length of ICU stay (Days)*** | 2.27 ± 0.83 | 8.59 ± 4.37 | 5.72 ± 6.13 | < 0.0011 |
| Mechanical ventilator (Required)*** | 0 (0.0%) | 77 (11.2%) | 243 (100.0%) | < 0.0012 |
| Number of co-morbidities*** | 3.26 ± 1.65 | 4.25 ± 1.58 | 6.51 ± 1.73 | < 0.0011 |
| Hypertension (Present)*** | 165 (44.5%) | 400 (58.3%) | 181 (74.5%) | < 0.0012 |
| Diabetes (Present) | 25 (6.7%) | 49 (7.1%) | 22 (9.1%) | 0.5292 |
| Renal disease (Present)*** | 95 (25.6%) | 254 (37.0%) | 168 (69.1%) | < 0.0012 |
| Neurological disease (Present)*** | 89 (24.0%) | 157 (22.9%) | 117 (48.1%) | < 0.0012 |
| Cardiovascular disease (Present)*** | 229 (61.7%) | 558 (81.3%) | 240 (98.8%) | < 0.0012 |
| Respiratory disease (Present)*** | 30 (8.1%) | 121 (17.6%) | 207 (85.2%) | < 0.0012 |
| Liver disease (Present)*** | 65 (17.5%) | 159 (23.2%) | 103 (42.4%) | < 0.0012 |
| Immunocompromised state (Present)*** | 73 (19.7%) | 255 (37.2%) | 75 (30.9%) | < 0.0012 |
| Hematological abnormality (Present)*** | 167 (45.0%) | 386 (56.3%) | 185 (76.1%) | < 0.0012 |
| Sepsis (Present)*** | 116 (31.3%) | 304 (44.3%) | 168 (69.1%) | < 0.0012 |
| Dyselectrolytemia (Present)*** | 50 (13.5%) | 151 (22.0%) | 97 (39.9%) | < 0.0012 |
| Other disorders (present)*** | 106 (28.7%) | 108 (15.7%) | 18 (7.4%) | < 0.0012 |
Abbreviations: APACGE, Acute Physiology and Chronic Health Evaluation; ICU, intensive care unit; PDW, platelet distribution width; RDW, red blood cell distribution width.
*** denotes statistically significant.
►Fig. 2 depicts the correlation between the length of ICU stay (days) and RDW (%). Each point depicts an individual case. The trendline in blue color shows the general trend of correlation between both variables. The 95% confidence interval of the above trendline is shaded in gray color. The correlation between both the variables was explored with the help of nonparametric test. The length of ICU stay and RDW (%), were moderately positively correlated and the correlation was statistically significant (rho = 0.37, p ≤ 0.001). For each unit increase in length of ICU stay (days), there was 0.09 units increase in RDW (%). Contrarily, for increment in each unit of RDW (%), the length of ICU stay (days) was increased by 0.25 units.
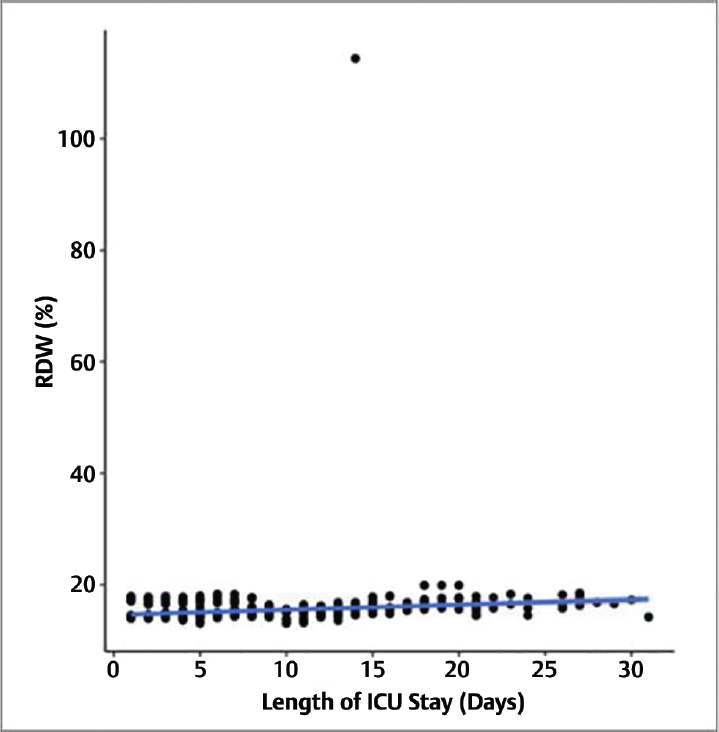
- Correlation between length of ICU stay (days) and RDW (%) (n = 1,300).
The area under the ROC curve (AUROC) for RDW (%) predicting outcome: death versus outcome: discharge was 0.966 (95% CI: 0.954–0.977), thus demonstrating excellent diagnostic performance. It was statistically significant (p ≤ 0.001) as evident in ►Fig. 3. At a cutoff of RDW (%) ≥ 15.9, it predicts outcome: death and the sensitivity was 88%, while specificity was 96%. The odds ratio (95% CI) for outcome: 1 when RDW (%) is ≥ 15.9 was 167.67 (103.5–271.61). The relative risk (95% CI) for outcome: 1 when RDW (%) is ≥ 15.9 was 26 (18.67–36.32).
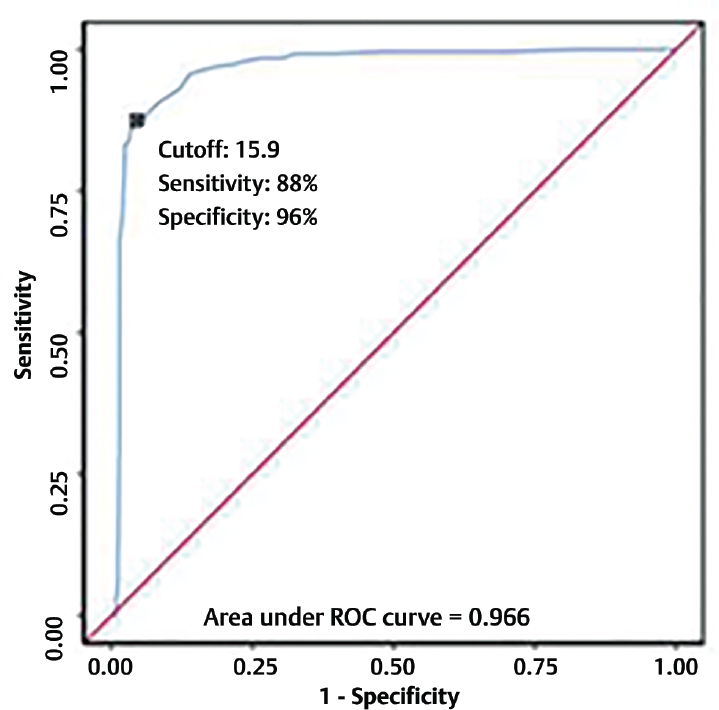
- ROC curve analysis showing diagnostic performance of RDW (%) in predicting outcome: death vs outcome discharge (n = 1,300).
►Fig. 4 depicts the correlation between the length of ICU stay (days) and PDW (%). Each point depicts an individual case. The trendline in blue color shows the general trend of correlation between both the variables. The 95% confidence interval of the above trendline is shaded in gray color. The correlation in between both the variables was explored with the help of nonparametric test. The length of ICU stay and PDW (%) were moderately positive correlated and the correlation was statistically significant (rho = 0.5, p ≤ 0.001) as evident in Fig. 4. For each unit increase in the length of ICU stay (days), there was 0.16 units increase in PDW (%). Contrarily, for increment in each unit of PDW (%), the length of ICU stay (days) increases by 0.71 units.

- Correlation between length of ICU stay (days) and PDW (%) (n = 1,300).
The AUROC for PDW (%) predicting outcome: death versus outcome: discharge was 0.973 (95% CI: 0.961–0.985), thus demonstrating excellent diagnostic performance. It was statistically significant (p ≤ 0.001) as evident in ►Fig. 5. At a cutoff of PDW (%) ≥ 14.1, it predicts outcome: death and sensitivity was 97%, while the specificity was 97%. The odds ratio (95% CI) for outcome: 1 when PDW (%) is ≤ 14.1 was 1009.91 (458.53–2224.31). The relative risk (95% CI) for outcome: 1 when PDW (%) is ≤ 14.1 was 113.5 (57.74–223.97).
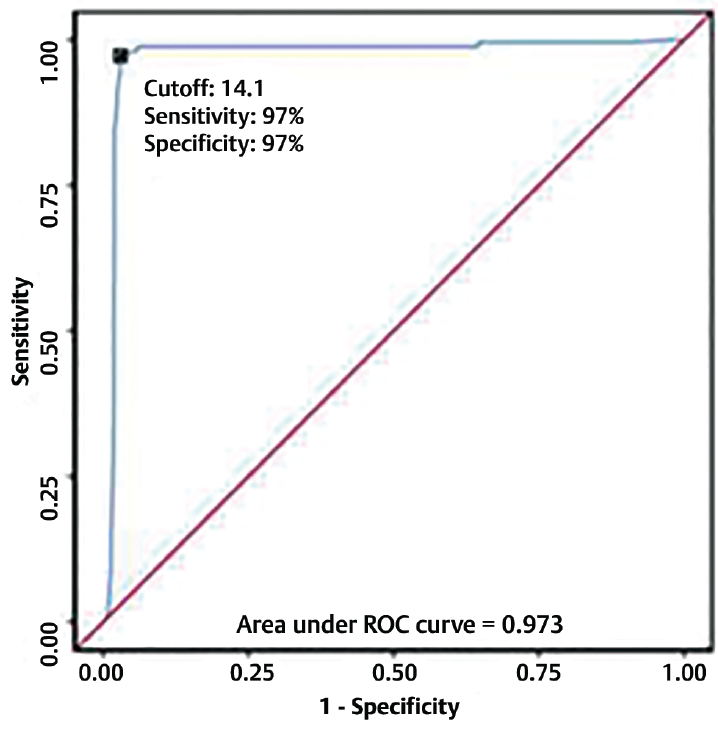
- ROC curve analysis showing diagnostic performance of PDW (%) in predicting outcome: death vs outcome discharge (n = 1,300).
APACHE IV score, PDW (%), and RDW (%) significantly predicted outcome: death, as evident in ►Fig. 6

- Comparison of the diagnostic performance of various predictors in predicting outcome: death vs outcome discharge (n = 1,300).
Discussion
The current study analyzed the impact of RDW and PDW in patients who were critically ill and admitted in MICU. The study predicted outcome in patients of MICUs with respect to the length of stay in ICU, the need for ventilator and death and compared RDW and PDW with APACHE IV score.
The mean age for the patients was 50.78 ± 17.51 years. 63.5% of the patients were males while 36.5% patients were females. In a study by Pal and Prashant[16]from Northeast India, authors audited reasons for mortality in patients admitted to MICUs. They noted that high number of patients with mortality had sepsis at 26.3%, stroke in 19.9% of the patients, and COPD in 13.9% of the patients. They noted that high number of deaths occurred in patients above 60 years. 10.5% of the patients had diabetes, 16.5% were hypertensive in this study. In another study by Madani et al[17] authors evaluated patterns of ICU admissions and mortality. Of 140 patients 63.6% had cardiovascular events and 26.4% had infections. The death rate was 10.7%.
The patterns of ICU admission might differ in different geographical areas and it would also differ based on specialty services provided. There can be a rural and urban variation considering differences in risk factors and underlying medical conditions. It should be noted that presence of multiple co-morbidities is associated with increased risk of longer duration of hospitalization, increased ICU stay, and mortality rate.[18]
The mean RDW (%) for the entire study population was 15.17 ± 3.01. The mean (SD) of RDW (%) in the age group of < 40 years was 14.81 ± 1.11, in age group of 40 to 60 years it was 15.27 ± 4.49, while in the age group > 60 years it was 15.34 ± 1.28. The median (IQR) of RDW (%) in patients with age group of < 40 years was 14.4 ± 0.9, in 40 to 60 years age group it was 14.6 ± 1.1, and in the age group of > 60 it was 14.9 ± 1.9. The RDW (%) in patient with age group of < 40 years ranged from 13.1 to 19.9, while in 40 to 60 years ranged from 13.2 to 14.4 and in age group of > 60 years ranged from 13.3 to 19.9. The difference in terms of RDW between the three age groups was significant (X2 = 43.725, p ≤ 0.001). The difference in gender distribution in terms of RDW (%) was not significant (U = 207431.000, p = 0.077). In a study by Hoffmann et al,[19] authors noted that there is an increase in RDW with increasing age, however, authors noted that this was not gender specific. This corresponds with our study.
The difference in both the groups (death vs. discharge) with regards to RDW (%) was significant (U = 9178.500, p ≤ 0.001), with the highest median RDW (%) in the outcome: death group. The mean (SD) of RDW (%) in those discharged was 14.72 ± 3.18 while those who died was 16.99 ± 0.80. The median (IQR) of RDW (%) in the outcome: discharge group was 14.4 ± 0.7, while in the outcome: death group was 17.2 ± 0.9. The RDW (%) in the outcome: discharge ranged from 13.1 to 14.4. while in the outcome: death it ranged from 14.2 to 18.3. The RDW (%) was remarkably higher in comparison with discharged patients. Various studies have noted that increase in RDW correlates with increase in mortality. Perlstein et al[20] in a study noted that increased RDW is a predictor of increased mortality. Şenol et al[21] noted that the mortality risk in acute pancreatitis patient is increased with increased RDW. Ani and Ovbiagele[22] noted that the mortality was increased in patients with stroke with increased RDW.
The correlation between the length of ICU stays (days) and RDW (%) was moderately positive and was significant (rho = 0.37, p ≤ 0.001). For each unit increase in the length of ICU stay (days), there is increment in the RDW (%) by 0.09 units. Contrarily, for increase in each unit of RDW (%), the length of ICU stay (days) increases by 0.25 units. Farzanegan and Zangi[23] noted that the increase in RDW is associated with longer duration of ICU stay. Tonietto et al[24] noted that elevated RDW is associated with increased chances for re-admission to ICU. Fujita et al[25] similarly noted a correlation between RDW and length of stay in ICU and hospitalization duration.
The mean (SD) of RDW (%) in the mechanical ventilator: required group was 17.07 ± 5.58 while in the mechanical ventilator: not required group was 14.55 ± 0.55. The median (IQR) of RDW (%) in the mechanical ventilator: required group was 17.15 ± 1.35 while in the mechanical ventilator: not required group was 14.4 ± 0.6. The RDW (%) in the mechanical ventilator: required group ranged from 13.8 to 14.4 while in the mechanical ventilator: not required group ranged from 13.1 to 16.8. The difference between both the groups with respect to RDW (%) was significant (U = 294104.500, p ≤ 0.001), with mechanical ventilator: required group having the highest median RDW (%). Otero et al[26] noted that increased RDW is associated with decreased ventilator free time in critically ill patients. Meynaar et al[27] similarly observed that increased RDW correlates with increased chances for intubation in patients who were critically ill.
The AUROC for RDW (%) predicting outcome: death versus outcome: discharge was 0.966 (95% CI: 0.954–0.977), thus demonstrating excellent diagnostic performance. It was statistically significant (p ≤ 0.001) as observed by various studies. RDW[20,21,22] is a promising prognostic factor in predicting death in patients requiring ICU support which correlated with our study.
The mean (SD) of PDW (%) in the age group of < 40 years was 10.93 ± 2.24 while in the 40 to 60 years age group it was 11.51 ± 2.31, and in the age group of > 60 years group it was 11.97 ± 2.53. The median (IQR) of PDW (%) in the age group of < 40 years was 10.2 ± 2.1, while in 40 to 60 years age group it was 10.5 ± 2.4. In > 60 years age group it was 10.9 ± 4.8. The PDW (%) in the age group < 40 years ranged from 9 to 17.1, while in the age group 40 to 60 years it ranged from 9 to 17.3, and in the age group of > 60 years it ranged from 9 to 17.4. The difference in terms of PDW (%) between the three age groups was significant (X2= 49.206, p ≤ 0.001), with the highest median PDW (%) in the age group > 60 years. The difference in gender distribution in terms of PDW (%) was not significant (U = 206445.000, p = 0.107). Use of PDW has been utilized in various workers in assessing prognosis in various diseases such as gastric cancer, appendicitis, thyroid disorders, etc.[28,29] Hoffmann and van den Broek[30] did not note any significant correlation between gender and platelet indices but however, noted age of the patient might have some association.
The mean (SD) of PDW (%) in the outcome: discharge group was 10.53 ± 1.48 while in the outcome: death group was 15.47 ± 0.87. The median (IQR) of PDW (%) in the outcome: discharge group was 10.2 ± 1.8 while in the outcome: death group was 15.6 ± 0.4. The PDW (%) in the outcome: discharge ranged from 9 to 17.4, while in the outcome: death ranged from 9.1 to 17.1. The difference between both the groups with respect to PDW (%) was significant (U = 7162.500, p ≤ 0.001), and the outcome: death group had the highest median PDW (%). Zhan et al[31] noted in a study that increase in PDW was noted in fatal cases when compared with the survivors. Simani et al[32] observed higher PDW values in patients who had a fatal course when suffering from post-neurosurgical meningitis when compared with the survivors. Gao et al[33] in a study on patients with septic shock noted higher chances of death in patients with increased PDW.
The correlation between length of ICU stay (days) and PDW (%) was moderately positive and was significant (rho = 0.5, p ≤ 0.001). For each unit increase in length of ICU stay (days), there was increment in PDW (%) by 0.16 units. Contrarily, for each unit increase in PDW (%), the length of ICU stay (days) increases by 0.71 units. Sezgi et al[34] observed in patients admitted to respiratory ICU that patients with higher value of PDW had longer duration of stay in ICU and higher mortality. Zhang et al[3] noted higher mean duration of ICU stay in critically ill Chinese patients when correlated with increased PDW.
The mean (SD) of PDW (%) in the mechanical ventilator: required group was 15.30 ± 1.03 while of mechanical ventilator: not required group was 10.26 ± 1.01. The median (IQR) of PDW (%) in the mechanical ventilator: required group was 15.5 ± 1. while in the mechanical ventilator: not required group was 10.2 ± 1.5. The PDW (%) in the mechanical ventilator: required group ranged from 10.4 to 17.4. The PDW (%) in the mechanical ventilator: not required group ranged from 9 to 13.7. The difference in between both the groups with respect to PDW (%) was significant (U = 312187.000, p ≤ 0.001), and the mechanical ventilator: required group had the highest median PDW (%). Tzur et al[35] noted increased chances of mechanical ventilation in patients with a higher mean PDW at the time of admission. Dundar et al[36] observed increased need for mechanical ventilation in patients with organophosphorus poisoning having elevated PDW.
The AUROC for PDW (%) in predicting outcome: death versus outcome: discharge was 0.973 (95% CI: 0.961–0.985), thus demonstrating excellent diagnostic performance. It was statistically significant (p ≤ 0.001). At a cut off of PDW [%] > 14.1, it predicts outcome: death with sensitivity 97% and specificity 97%. PDW, as observed from our study, can be an important prognostic marker in patients who are admitted in ICUs.
In APACHE IV, the diagnosis on admission to the ICU, days of stay in the hospital before ICU care, thrombolytic therapy, and mechanical ventilation are added and this is uncommonly used. Higher scores of APACHE IV are associated with more critical illness with increased chances of ventilation, increased ICU stay, increased hospitalization, and mortality.[37] Considering higher scores of APACHE IV with increased levels of PDW, the scoring system corresponds with our findings related to RDW and PDW being markers for increased ICU stay, increased need for mechanical ventilation, and increased mortality.
Limitations
The study population was nonhomogeneous. As such confounding effect of underlying medical conditions such as causes of sepsis like chronic kidney disease, liver disease, ARDS on mortality was not studied and cannot be totally excluded. The study did not evaluate other platelet and red cells indices. Serial, multiple PDW and RDW estimations and its effect on the clinical outcomes were not done.
Conclusion
RDW and PDW were found as significant indicators for period of stay in ICU, requirement for mechanical ventilation, and mortality rate in patients admitted to ICUs. The indices also correlated with APACHE IV scoring system, with higher RDW and PDW values in patients leading to a higher APACHE IV score, which in turn correlated with the period of stay in ICU, requirement for mechanical ventilation, and mortality rate. As these are simple, easy to conduct, and universally available tests, they can be regularly incorporated in patients admitted to ICUs. Adequate interventions can also be planned based on these parameters for better patient outcomes.
Conflict of Interest
None declared.
References
- Red blood cell distribution width (RDW) and human pathology. One size fits all. Clin Chem Lab Med. 2014;52(09):1247-1249.
- [CrossRef] [Google Scholar]
- et al Platelet size deviation width, platelet large cell ratio, and mean platelet volume have sufficient sensitivity and specificity in the diagnosis of immune thrombocytopenia. Br J Haematol. 2005;128(05):698-702.
- [CrossRef] [PubMed] [Google Scholar]
- Use of platelet indices for determining illness severity and predicting prognosis in critically ill patients. Chin Med J (Engl). 2015;128(15):2012-2018.
- [CrossRef] [PubMed] [Google Scholar]
- The role of red blood cell distribution width in cardiovascular and thrombotic disorders. Clin Chem Lab Med. 2011;50(04):635-641.
- [CrossRef] [Google Scholar]
- Relation between red cell distribution width and clinical outcomes after acute myocardial infarction. Am J Cardiol. 2010;105(03):312-317.
- [CrossRef] [PubMed] [Google Scholar]
- et al Red blood cell distribution width predicts long-term outcome regardless of anaemia status in acute heart failure patients. Eur J Heart Fail. 2009;11(09):840-846.
- [CrossRef] [PubMed] [Google Scholar]
- Elevated red cell distribution width predicts poor outcome in young patients with community acquired pneumonia. Crit Care. 2011;15(04):R194.
- [CrossRef] [PubMed] [Google Scholar]
- Usefulness of red cell distribution width as a prognostic marker in pulmonary hypertension. Am J Cardiol. 2009;104(06):868-872.
- [CrossRef] [PubMed] [Google Scholar]
- Red blood cell distribution width and the risk of death in middle-aged and older adults. Arch Intern Med. 2009;169(05):515-523.
- [CrossRef] [PubMed] [Google Scholar]
- Red cell distribution width and all-cause mortality in critically ill patients. Crit Care Med. 2011;39(08):1913-1921.
- [CrossRef] [PubMed] [Google Scholar]
- Platelet large cell ratio in the differential diagnosis of abnormal platelet counts. Indian J Pathol Microbiol. 2004;47(02):202-205.
- [Google Scholar]
- Platelet distribution width (PDW): a putative marker for threatened preterm labour. Pak J Med Sci. 2014;30(04):745-748.
- [CrossRef] [Google Scholar]
- et al Premature labor: a state of platelet activation? J Perinat Med. 2008;36(05):377-387.
- [CrossRef] [Google Scholar]
- et al Association between PDW and long term major adverse cardiac events in patients with acute coronary syndrome. Heart Lung Circ. 2016;25(01):29-34.
- [CrossRef] [PubMed] [Google Scholar]
- Platelet counts, MPV and PDW in culture proven and probable neonatal sepsis and association of platelet counts with mortality rate. J Coll Physicians Surg Pak. 2014;24(05):340-344.
- [Google Scholar]
- An audit on pattern of mortality in a medicine ICU at tertiary care hospital of North-East India. Int J Med Biomed Stud. 2020;4(02):199-201.
- [CrossRef] [Google Scholar]
- et al Causes of admission to intensive care units in the Hajj period of the Islamic year 1424 (2004) Ann Saudi Med. 2007;27(02):101-105.
- [CrossRef] [PubMed] [Google Scholar]
- Multiple cardiovascular comorbidities and acute myocardial infarction: temporal trends (1990-2007) and impact on death rates at 30 days and 1 year. Clin Epidemiol. 2012;4:115-123.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of age and gender on reference intervals of red blood cell distribution width (RDW) and mean red cell volume (MCV) Clin Chem Lab Med. 2015;53(12):2015-2019.
- [CrossRef] [Google Scholar]
- Red blood cell distribution width and mortality risk in a community-based prospective cohort. Arch Intern Med. 2009;169(06):588-594.
- [CrossRef] [PubMed] [Google Scholar]
- Red cell distribution width as a predictor of mortality in acute pancreatitis. Am J Emerg Med. 2013;31(04):687-689.
- [CrossRef] [PubMed] [Google Scholar]
- Elevated red blood cell distribution width predicts mortality in persons with known stroke. J Neurol Sci. 2009;277(1-2):103-108.
- [CrossRef] [PubMed] [Google Scholar]
- Predictor factors for sepsis diagnosis, length of ICU stay and mortality in ICU. J Cell Mol Anesth. 2017;2(02):55-62.
- [Google Scholar]
- et al Elevated red blood cell distribution width at ICU discharge is associated with readmission to the intensive care unit. Clin Biochem. 2018;55:15-20.
- [CrossRef] [PubMed] [Google Scholar]
- et al Altered red blood cell distribution width in overweight adolescents and its association with markers of inflammation. Pediatr Obes. 2013;8(05):385-391.
- [CrossRef] [PubMed] [Google Scholar]
- Elevated red cell distribution width at initiation of critical care is associated with mortality in surgical intensive care unit patients. J Crit Care. 2016;34:7-11.
- [CrossRef] [PubMed] [Google Scholar]
- et al Red cell distribution width as predictor for mortality in critically ill patients. Neth J Med. 2013;71(09):488-493.
- [Google Scholar]
- et al The analysis of mean platelet volume and platelet distribution width levels in appendicitis. Indian J Surg. 2015;77(Suppl. 02):495-500.
- [CrossRef] [PubMed] [Google Scholar]
- et al No associations exist between mean platelet volume or platelet distribution width and thyroid function in Chinese. Medicine (Baltimore). 2016;95(40):e4573.
- [CrossRef] [PubMed] [Google Scholar]
- Reference intervals of reticulated platelets and other platelet parameters and their associations. Arch Pathol Lab Med. 2013;137(11):1635-1640.
- [CrossRef] [PubMed] [Google Scholar]
- Platelet indices are novel predictors of hospital mortality in intensive care unit patients. J Crit Care. 2014;29(05):885.e1-885.e6.
- [CrossRef] [PubMed] [Google Scholar]
- et al Platelet indices for predicting patient outcomes in post-neurosurgical meningitis. Arch Neurosci. 2019;6(02):e82911.
- [CrossRef] [Google Scholar]
- et al The impact of various platelet indices as prognostic markers of septic shock. PLoS ONE. 2014;9(08):e103761.
- [CrossRef] [PubMed] [Google Scholar]
- et al Alterations in platelet count and mean platelet volume as predictors of patient outcome in the respiratory intensive care unit. Clin Respir J. 2015;9(04):403-408.
- [CrossRef] [PubMed] [Google Scholar]
- et al Platelet distribution width: a novel prognostic marker in an internal medicine ward. J Community Hosp Intern Med Perspect. 2019;9(06):464-470.
- [CrossRef] [PubMed] [Google Scholar]
- Prognostic value of red cell distribution width in patients with organophosphate poisoning. J Acad Emerg Med. 2015;14(02):65-69.
- [CrossRef] [Google Scholar]
- Acute Physiology and Chronic Health Evaluation (APACHE) IV: hospital mortality assessment for today's critically ill patients. Crit Care Med. 2006;34(05):1297-1310.
- [CrossRef] [PubMed] [Google Scholar]







